| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2018-05-02 12:54:50 UTC |
|---|
| Update date | 2018-05-04 14:12:50 UTC |
|---|
| FoodComEx ID | PC001221 |
|---|
| FoodDB Record | FDB000918 |
|---|
| Chemical Information |
|---|
| Name | Saccharin |
|---|
| Description | Widely-used sweetening agent. All salts intensely sweet. Permitted in foods at levels of 80-1200 ppm in EU
Although saccharin was commercialized not long after its discovery, it was not until sugar shortages during World War I that its use became widespread. Its popularity further increased during the 1960s and 1970s among dieters, since saccharin is a calorie-free sweetener. In the United States saccharin is often found in restaurants in pink packets; the most popular brand is "Sweet'N Low". A small number of soft drinks are sweetened with saccharin, the most popular[citation needed] being the Coca-Cola Company's cola drink Tab, introduced in 1963 as a diet cola soft drink.; Harvey Wiley was one particularly well-known figure involved in the investigation of saccharin. Wiley, then the director of the bureau of chemistry for the USDA, had suspected saccharin to be damaging to human health. Wiley first battled saccharin in 1908. In a clash that epitomizes the controversial history of saccharin, Harvey told then President Theodore Roosevelt directly that "Everyone who ate that sweet corn was deceived. He thought he was eating sugar, when in point of fact he was eating a coal tar product totally devoid of food value and extremely injurious to health." In a heated exchange, Roosevelt angrily answered Harvey by stating "Anybody who says saccharin is injurious to health is an idiot." In 1911, the Food Inspection Decision 135 stated that foods containing saccharin were adulterated. However in 1912, Food Inspection Decision 142 stated that saccharin was not harmful. The government's stance on saccharin has continued to waver ever since. More controversy was stirred in 1969 with the discovery of files from the Food and Drug Administration's investigations of 1948 and 1949. These investigations, which had originally argued against saccharin use, were shown to prove little about saccharin being harmful to human health. In 1972 the USDA made an attempt to completely ban the substance. However, this attempt was unsuccessful and the sweetener remains widely available in the United States; it is the third-most popular after sucralose and aspartame. Cyclamate, however, was banned in the US and saccharin was banned in Canada, leading to different product formulations being marketed in these countries.; Saccharin has the chemical formula C7H5NO3S and it can be produced in various ways. The original route starts with toluene, but yields from this starting point are low. In 1950, an improved synthesis was developed at the Maumee Chemical Company of Toledo, Ohio. In this synthesis, anthranilic acid successively reacts with nitrous acid, sulfur dioxide, chlorine, and then ammonia to yield saccharin. Another route begins with o-chlorotoluene. It is also known as ortho sulfobenzoic acid.; Saccharin is an acid with a pKa of about 2.; Saccharin is an artificial sweetener. The basic substance, benzoic sulfinide, has effectively no food energy and is much sweeter than sucrose, but has an unpleasant bitter or metallic aftertaste, especially at high concentrations. In countries where saccharin is allowed as a food additive, it is used to sweeten products such as drinks, candies, medicines, and toothpaste.; Saccharin was first produced in 1878 by Constantin Fahlberg, a chemist working on coal tar derivatives in Ira Remsen's laboratory at the Johns Hopkins University. It was Fahlberg who, accidentally, discovered its intensely sweet nature.[citation needed] Fahlberg and Remsen published articles on benzoic sulfinide in 1879 and 1880. In 1884, now working on his own in New York City, Fahlberg applied for patents in several countries describing methods of producing this substance that he named saccharin. Fahlberg would soon grow wealthy, while Remsen merely grew irate, believing that he deserved credit for substances produced in his laboratory. On the matter, Remsen commented, "Fahlberg is a scoundrel. It nauseates me to hear my name mentioned in the same breath with him.". |
|---|
| CAS Number | 81-07-2 |
|---|
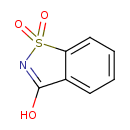
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| 1, 2-Benzisothiazol-3(2H)-one, 1,1-dioxide | biospider | | 1, 2-Benzisothiazolin-3-one 1,1-dioxide | biospider | | 1, 2-Dihydro-2-ketobenzisosulfonazole | biospider | | 1,1-Diox-1,2-benzisothiazol-3-one | biospider | | 1,1-Dioxide-1,2-benzisothiazol-3(2H)-one | biospider | | 1,1-Dioxide-1,2-benzisothiazolin-3-one | biospider | | 1,1-Dioxo-1,2-benzisothiazol-3(2H)-one | biospider | | 1,1-Dioxo-1,2-dihydro-1lambda*6*-benzo[d]isothiazol-3-one | biospider | | 1,1-Dioxo-1,2-dihydro-benzo[d]isothiazol-3-one | biospider | | 1,2-Benzisothiazol-3(2H)-one 1,1-dioxide, 9CI | db_source | | 1,2-Benzisothiazol-3(2H)-one, 1,1-dioxide | biospider | | 1,2-Benzisothiazolin-3-one 1,1-dioxide | biospider | | 1,2-Benzisothiazolin-3-one, 1,1-dioxide | biospider | | 1,2-Benzisothiazoline-3-one 1,1-dioxide | biospider | | 1,2-Benzothiazol-3(2H)-one 1,1-dioxide | biospider | | 1,2-Dihydro-2-ketobenzisosulfonazole | biospider | | 1,2-Dihydro-2-ketobenzisosulphonazole | biospider | | 2-Sulfobenzoic acid imide | biospider | | 2-Sulfobenzoicimide | biospider | | 2-Sulphobenzoic imide | biospider | | 2, 3-Dihydro-3-oxobenzisosulfonazole | biospider | | 2,3-Dihydro-1,2-benzoisothiazol-3-one-1,1-dioxide | biospider | | 2,3-Dihydro-3-oxobenzisosulfonazole | biospider | | 2,3-Dihydro-3-oxobenzisosulphonazole | biospider | | 2,3-Dihydroxy-1,2-benzisothiazol-3-one-1,1-dioxide | biospider | | 3-Benzisothiazolinone 1, 1-dioxide | biospider | | 3-Benzisothiazolinone 1,1-dioxide | biospider | | 3-Hydroxybenzisothiazole S,S-dioxide | biospider | | 3-Hydroxybenzisothiazole-S,S-dioxide | biospider | | Anhydro-o-sulfaminebenzoic acid | biospider | | Benzisosulfonazole, 2,3-dihydro-3-oxo- | biospider | | Benzo-2-sulfiide | biospider | | Benzo-2-sulphimide | biospider | | Benzo-sulphinide | biospider | | Benzoic acid sulfimide | biospider | | Benzoic sulfimide | db_source | | Benzoic sulphimide | biospider | | Benzosulfimide | biospider | | Benzosulfimide, o- | biospider | | Benzosulfinide | db_source | | Benzosulphimide | biospider | | Benzoylsulfonic imide | biospider | | E954 | db_source | | Garantose | db_source | | Glucid | biospider | | Gluside | biospider | | Glycophenol | biospider | | Hermesetas | db_source | | Insoluble saccharin | biospider | | Kandiset | biospider | | LSA | biospider | | Neosaccharin | biospider | | O-benzoic acid sulfimide | biospider | | O-benzoic sulfimide | biospider | | O-benzoic sulphimide | biospider | | o-Benzosulfimide | db_source | | O-benzosulphimide | biospider | | O-benzoyl sulfimide | biospider | | O-benzoyl sulphimide | biospider | | o-Benzoylsulfimide | db_source | | O-sulfobenzimide | biospider | | O-sulfobenzoic acid imide | biospider | | o-Sulfobenzoic imide | db_source | | Sacarina | biospider | | Saccharimide | biospider | | Saccharin (JP15/NF) | biospider | | Saccharin [usan] | biospider | | Saccharin acid | biospider | | Saccharin insoluble | biospider | | Saccharin, insoluble | biospider | | Saccharine | biospider | | Saccharinol | biospider | | Saccharinose | biospider | | Saccharol | biospider | | Sacharin | biospider | | Stilalgin | biospider | | Sucre edulcor | biospider | | Sucrette | biospider | | Sulfobenzimide, o- | biospider | | Sweeta | db_source | | Syncal | biospider | | Zaharina | biospider |
|
|---|
| Chemical Formula | C7H5NO3S |
|---|
| IUPAC name | 2,3-dihydro-1lambda6,2-benzothiazole-1,1,3-trione |
|---|
| InChI Identifier | InChI=1S/C7H5NO3S/c9-7-5-3-1-2-4-6(5)12(10,11)8-7/h1-4H,(H,8,9) |
|---|
| InChI Key | CVHZOJJKTDOEJC-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | O=C1NS(=O)(=O)C2=C1C=CC=C2 |
|---|
| Average Molecular Weight | 183.185 |
|---|
| Monoisotopic Molecular Weight | 182.999013721 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as benzothiazoles. These are organic compounds containing a benzene fused to a thiazole ring (a five-membered ring with four carbon atoms, one nitrogen atom and one sulfur atom). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Benzothiazoles |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Benzothiazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1,2-benzothiazole
- Benzenoid
- Organosulfonic acid or derivatives
- Organic sulfonic acid or derivatives
- Azacycle
- Carboxylic acid derivative
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | 0.91 | HANSCH,C ET AL. (1995) |
|---|
| Experimental Water Solubility | 4 mg/mL at 25 oC | SEIDELL,A (1941) |
|---|
| Melting Point | Mp 224° | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | 1 to 2 mg |
|---|
| Delivery Time | 2 weeks |
|---|
| Storage Form | powder |
|---|
| Storage Conditions | -18°C |
|---|
| Stability | Not Available |
|---|
| Purity | unknown |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
| Contact Name | Contact Institution | Contact Email |
|---|
| Augustin Scalbert | International Agency for Research on Cancer (IARC), Biomarkers Group, 150 cours Albert Thomas, Lyon, FR, 69372 | scalberta@iarc.fr |
|
| Commercial Vendors |
|---|
| AKSci | Z1771 |
|---|
| Glentham | GX2610 |
|---|
| Toronto Research Chemicals | S080800 |
|---|