| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2018-05-02 11:57:37 UTC |
|---|
| Update date | 2018-05-04 14:25:57 UTC |
|---|
| FoodComEx ID | PC001170 |
|---|
| FoodDB Record | FDB001978 |
|---|
| Chemical Information |
|---|
| Name | 7-Methylxanthine |
|---|
| Description | 7-Methylxanthine, also known as heteroxanthin, belongs to the class of organic compounds known as xanthines. These are purine derivatives with a ketone group conjugated at carbons 2 and 6 of the purine moiety. 7-Methylxanthine is an extremely weak basic (essentially neutral) compound (based on its pKa). 7-Methylxanthine exists in all living organisms, ranging from bacteria to humans. Within humans, 7-methylxanthine participates in a number of enzymatic reactions. In particular, 7-methylxanthine and formaldehyde can be biosynthesized from paraxanthine through the action of the enzyme cytochrome P450 1A2. In addition, 7-methylxanthine can be converted into 7-methyluric acid through the action of the enzyme xanthine dehydrogenase/oxidase. In humans, 7-methylxanthine is involved in caffeine metabolism. Outside of the human body, 7-Methylxanthine has been detected, but not quantified in, arabica coffee. This could make 7-methylxanthine a potential biomarker for the consumption of these foods. |
|---|
| CAS Number | 552-62-5 |
|---|
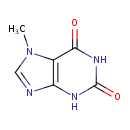
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| 2,6-Dihydroxy-7-methylpurine | HMDB | | 3,7-dihydro-7-Methyl-1H-purine-2,6-dione | ChEBI | | 3,7-Dihydro-7-methyl-1H-purine-2,6-dione, 9CI | db_source | | 7-Methyl-3,7-dihydro-1H-purine-2,6-dione | HMDB | | 7-Methyl-7H-purine-2,6-diol | HMDB | | 7-Methylxanthin | ChEBI | | 7-Methylxanthine | db_source | | Heteroxanthin | ChEBI | | Heteroxanthine | db_source | | Methylxanthine | HMDB |
|
|---|
| Chemical Formula | C6H6N4O2 |
|---|
| IUPAC name | 7-methyl-2,3,6,7-tetrahydro-1H-purine-2,6-dione |
|---|
| InChI Identifier | InChI=1S/C6H6N4O2/c1-10-2-7-4-3(10)5(11)9-6(12)8-4/h2H,1H3,(H2,8,9,11,12) |
|---|
| InChI Key | PFWLFWPASULGAN-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | CN1C=NC2=C1C(=O)NC(=O)N2 |
|---|
| Average Molecular Weight | 166.1374 |
|---|
| Monoisotopic Molecular Weight | 166.049075456 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as xanthines. These are purine derivatives with a ketone group conjugated at carbons 2 and 6 of the purine moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Imidazopyrimidines |
|---|
| Sub Class | Purines and purine derivatives |
|---|
| Direct Parent | Xanthines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Xanthine
- Alkaloid or derivatives
- Hydroxypyrimidine
- Pyrimidine
- N-substituted imidazole
- Heteroaromatic compound
- Imidazole
- Azole
- Azacycle
- Hydrocarbon derivative
- Organic oxygen compound
- Organic nitrogen compound
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | -0.89 | GASPARI,F & BONATI,M (1987) |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Mp 380° dec. | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | 1 to 2 mg |
|---|
| Delivery Time | 2 weeks |
|---|
| Storage Form | powder |
|---|
| Storage Conditions | -18°C |
|---|
| Stability | Not Available |
|---|
| Purity | unknown |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
| Contact Name | Contact Institution | Contact Email |
|---|
| Augustin Scalbert | International Agency for Research on Cancer (IARC), Biomarkers Group, 150 cours Albert Thomas, Lyon, FR, 69372 | scalberta@iarc.fr |
|
| Commercial Vendors |
|---|
| AKSci | 8053AB |
|---|
| MetaSci | HMDB0001991 |
|---|
| Sigma-Aldrich | HMDB0001991 |
|---|
| Toronto Research Chemicals | M338580 |
|---|