| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:33:32 UTC |
|---|
| Update date | 2017-01-19 02:36:42 UTC |
|---|
| FoodComEx ID | PC000943 |
|---|
| FoodDB Record | FDB001089 |
|---|
| Chemical Information |
|---|
| Name | (S)-N-Acetylmethionine |
|---|
| Description | Nutrient supplement used as a source of L-methionine. |
|---|
| CAS Number | 65-82-7 |
|---|
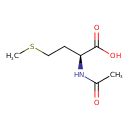
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| Acetyl-L-Methionine | biospider | | Acetylmethionin | HMDB | | Acetylmethionine | biospider | | DL-N-Acetylmethionine | HMDB | | L-(N-Acetyl)methionine | biospider | | L-Methionine, N-acetyl- | biospider | | L-N-Acetyl-methionine | biospider | | Methionamine | biospider | | Methionin | HMDB | | Methionine, N-acetyl- | biospider | | Methionine, N-acetyl-, L- (8CI) | biospider | | N-Acetyl-L-methionine | biospider | | N-Acetyl-methionine | biospider | | N-Acetyl-S-methylhomocysteine | biospider | | N-Acetyl(methyl)homocysteine | HMDB | | N-Acetylmethionine | biospider | | Thiomedon | HMDB |
|
|---|
| Chemical Formula | C7H13NO3S |
|---|
| IUPAC name | (2S)-2-acetamido-4-(methylsulfanyl)butanoic acid |
|---|
| InChI Identifier | InChI=1S/C7H13NO3S/c1-5(9)8-6(7(10)11)3-4-12-2/h6H,3-4H2,1-2H3,(H,8,9)(H,10,11)/t6-/m0/s1 |
|---|
| InChI Key | XUYPXLNMDZIRQH-LURJTMIESA-N |
|---|
| Isomeric SMILES | [H][C@@](CCSC)(NC(C)=O)C(O)=O |
|---|
| Average Molecular Weight | 191.248 |
|---|
| Monoisotopic Molecular Weight | 191.061613977 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as methionine and derivatives. Methionine and derivatives are compounds containing methionine or a derivative thereof resulting from reaction of methionine at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Methionine and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Methionine or derivatives
- N-acyl-alpha-amino acid
- N-acyl-alpha amino acid or derivatives
- N-acyl-l-alpha-amino acid
- Thia fatty acid
- Fatty acid
- Fatty acyl
- Acetamide
- Secondary carboxylic acid amide
- Carboxamide group
- Dialkylthioether
- Sulfenyl compound
- Thioether
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Hydrocarbon derivative
- Organic oxygen compound
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Carbonyl group
- Organic oxide
- Organopnictogen compound
- Organic nitrogen compound
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | -0.03 | MEYLAN,WM & HOWARD,PH (1995) |
|---|
| Experimental Water Solubility | 307 mg/mL at 25 oC | BEILSTEIN |
|---|
| Melting Point | Mp 104° | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 3 g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | F966 |
|---|
| AKSci | J92115 |
|---|
| Glentham | GM6577 |
|---|
| MetaSci | HMDB0011745 |
|---|
| Sigma-Aldrich | HMDB0011745 |
|---|