| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:33:18 UTC |
|---|
| Update date | 2017-01-19 02:36:40 UTC |
|---|
| FoodComEx ID | PC000899 |
|---|
| FoodDB Record | FDB022625 |
|---|
| Chemical Information |
|---|
| Name | Prostaglandin E1 |
|---|
| Description | Prostaglandin E1 (PGE1) is a potent endogenous vasodilator agent that increases peripheral blood flow. It inhibits platelet aggregation and has many other biological effects such as bronchodilation, mediation of inflammation, and various protective functions.
The protective action of PGE1 has been shown on both experimental animal models of liver injury and patients with fulminant viral hepatitis. PGE1-treated cirrhotic rats had less hepatosplenomegaly, lower serum alanine aminotransferase levels, and portal pressures and higher arterial pressure than placebo-treated cirrhotic rats.
Mechanisms of hepatic cytopotection of PGE1: inhibitory effects on T-cell mediated cytotoxicity, enhance DNA synthesis of injured liver after partial hepatectomy by stimulating cyclic AMP production and increasing ATP level in hepatic tissue, and could accelerate the recovery of mitochondrial respiratory functio n after reperfusion, stabilization of membrane microviscosity.
PGE1 is a prostanoid. Prostanoids is a term that collectively describes prostaglandins, prostacyclines and thromboxanes. Prostanoids are a subclass of the lipid mediator group known as eicosanoids. They derive from C-20 polyunsaturated fatty acids, mainly dihomo-gamma-linoleic (20:3n-6), arachidonic (20:4n-6), and eicosapentaenoic (20:5n-3) acids, through the action of cyclooxygenases-1 and -2 (COX-1 and COX-2). (PMID: 11819590, 16986207)
Prostaglandins are eicosanoids. The eicosanoids consist of the prostaglandins (PGs), thromboxanes (TXs), leukotrienes (LTs) and lipoxins (LXs). The PGs and TXs are collectively identified as prostanoids. Prostaglandins were originally shown to be synthesized in the prostate gland, thromboxanes from platelets (thrombocytes) and leukotrienes from leukocytes, hence the derivation of their names. All mammalian cells except erythrocytes synthesize eicosanoids. These molecules are extremely potent, able to cause profound physiological effects at very dilute concentrations. All eicosanoids function locally at the site of synthesis, through receptor-mediated G-protein linked signaling pathways. [HMDB] |
|---|
| CAS Number | 745-65-3 |
|---|
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (-)-3-hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-Cyclopentaneheptanoate | hmdb | | (-)-3-hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-Cyclopentaneheptanoic acid | hmdb | | (-)-Prostaglandin E1 | hmdb | | (+)-3-hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-Cyclopentaneheptanoate | hmdb | | (+)-3-hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-Cyclopentaneheptanoic acid | hmdb | | (11a,13e,15S)-11,15-Dihydroxy-9-oxoprost-13-en-1-Oate | Generator | | (11a,13e,15S)-11,15-Dihydroxy-9-oxoprost-13-en-1-Oic acid | Generator | | (11alpha,13e,15S)-11,15-Dihydroxy-9-oxoprost-13-en-1-Oate | Generator | | (11alpha,13e,15S)-11,15-Dihydroxy-9-oxoprost-13-en-1-Oic acid | ChEBI | | (11α,13e,15S)-11,15-dihydroxy-9-oxoprost-13-en-1-Oate | Generator | | (11α,13e,15S)-11,15-dihydroxy-9-oxoprost-13-en-1-Oic acid | Generator | | (13E)-(15S)-11-alpha,15-dihydroxy-9-oxoprost-13-enoate | hmdb | | (13E)-(15S)-11-alpha,15-dihydroxy-9-oxoprost-13-enoic acid | hmdb | | (13E)-(15S)-11,15-dihydroxy-9-oxoprost-13-enoate | hmdb | | (13E)-(15S)-11,15-dihydroxy-9-oxoprost-13-enoic acid | hmdb | | (13e)-(15S)-11a,15-Dihydroxy-9-oxoprost-13-enoate | Generator | | (13e)-(15S)-11a,15-Dihydroxy-9-oxoprost-13-enoic acid | Generator | | (13e)-(15s)-11alpha,15-dihydroxy-9-oxoprost-13-enoate | hmdb | | (13e)-(15s)-11alpha,15-dihydroxy-9-oxoprost-13-enoic acid | hmdb | | (13e)-(15S)-11α,15-dihydroxy-9-oxoprost-13-enoate | Generator | | (13e)-(15S)-11α,15-dihydroxy-9-oxoprost-13-enoic acid | Generator | | 11,15-Dihydroxy-9-oxoprost-13-en-1-oate | hmdb | | 11,15-Dihydroxy-9-oxoprost-13-en-1-oic acid | hmdb | | 11,15-dihydroxy-9-oxoprost-13-en-1-oic acid (ACD/Name 4.0) | hmdb | | 11a,15a-Dihydroxy-9-oxo-13-trans-prostenoate | Generator | | 11a,15a-Dihydroxy-9-oxo-13-trans-prostenoic acid | Generator | | 11alpha,15alpha-Dihydroxy-9-oxo-13-trans-prostenoate | Generator | | 11alpha,15alpha-Dihydroxy-9-oxo-13-trans-prostenoic acid | ChEBI | | 11α,15α-dihydroxy-9-oxo-13-trans-prostenoate | Generator | | 11α,15α-dihydroxy-9-oxo-13-trans-prostenoic acid | Generator | | 3-Hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-cyclopentaneheptanoate | hmdb | | 3-Hydroxy-2-(3-hydroxy-1-octenyl)-5-oxo-cyclopentaneheptanoic acid | hmdb | | Alprostadil | hmdb | | Alprostadil Prostoglandin E1 | hmdb | | Alprostadil(usan) | hmdb | | Alprostadilum | ChEBI | | Befar | hmdb | | Befar (TN) | hmdb | | Caverject | hmdb | | EDEX | hmdb | | l-Prostaglandin E1 | hmdb | | MUSE | hmdb | | PGE-1 | ChEBI | | PGE1 | hmdb | | Prink | hmdb | | Prink (TN) | hmdb | | prostaglandin E1 | hmdb | | Prostin VR | hmdb |
|
|---|
| Chemical Formula | C20H34O5 |
|---|
| IUPAC name | 7-[(1R,2R,3R)-3-hydroxy-2-[(3S)-3-hydroxyoct-1-en-1-yl]-5-oxocyclopentyl]heptanoic acid |
|---|
| InChI Identifier | InChI=1S/C20H34O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h12-13,15-17,19,21,23H,2-11,14H2,1H3,(H,24,25)/t15-,16+,17+,19+/m0/s1 |
|---|
| InChI Key | GMVPRGQOIOIIMI-DODZYUBVSA-N |
|---|
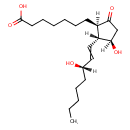
| Isomeric SMILES | CCCCC[C@H](O)C=C[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O |
|---|
| Average Molecular Weight | 354.481 |
|---|
| Monoisotopic Molecular Weight | 354.240624198 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as prostaglandins and related compounds. These are unsaturated carboxylic acids consisting of a 20 carbon skeleton that also contains a five member ring, and are based upon the fatty acid arachidonic acid. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Fatty Acyls |
|---|
| Sub Class | Eicosanoids |
|---|
| Direct Parent | Prostaglandins and related compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - Prostaglandin skeleton
- Long-chain fatty acid
- Fatty alcohol
- Hydroxy fatty acid
- Cyclopentanol
- Cyclic alcohol
- Cyclic ketone
- Ketone
- Secondary alcohol
- Carboxylic acid
- Carboxylic acid derivative
- Monocarboxylic acid or derivatives
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Carbonyl group
- Organooxygen compound
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework | Aliphatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 10 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | K274 |
|---|
| AKSci | SYN3025 |
|---|
| Cayman Chemical | 13010 |
|---|
| Toronto Research Chemicals | P838600 |
|---|