| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:33:15 UTC |
|---|
| Update date | 2017-01-19 02:36:40 UTC |
|---|
| FoodComEx ID | PC000893 |
|---|
| FoodDB Record | FDB022986 |
|---|
| Chemical Information |
|---|
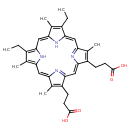
| Name | Mesoporphyrin IX |
|---|
| Description | Mesoporphyrins are porphyrins with four methyl, two ethyl, and two propionic acid side chains attached to the pyrrole rings. There are fifteen isomers of mesoporphyrin.
Mesoporphyrin has anti-inflammatory action. Mesoporphyrin inhibits interferon-gamma and interleukin-6 production, which is closely related to the suppression of prostaglandin E2 generation by interfering cyclooxygenase 1 and 2 enzyme activities suggesting that the inhibition of cytokine production is one of the anti-inflammatory mechanisms of mesoporphyrin. (PMID: 10446754) [HMDB] |
|---|
| CAS Number | 493-90-3 |
|---|
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| 7,12-diethyl-3,8,13,17-tetramethyl-2,18-Porphinedipropionic acid | hmdb | | 7,12-diethyl-3,8,13,17-tetramethyl-21H,23H-Porphine-2,18-dipropanoate | hmdb | | 7,12-diethyl-3,8,13,17-tetramethyl-21H,23H-Porphine-2,18-dipropanoic acid | hmdb | | Mesoporphyrin | hmdb | | Mesoporphyrin IX | hmdb | | MPIX | hmdb |
|
|---|
| Chemical Formula | C34H38N4O4 |
|---|
| IUPAC name | 3-[20-(2-carboxyethyl)-10,15-diethyl-5,9,14,19-tetramethyl-21,22,23,24-tetraazapentacyclo[16.2.1.1³,⁶.1⁸,¹¹.1¹³,¹⁶]tetracosa-1,3(24),4,6,8,10,12,14,16,18(21),19-undecaen-4-yl]propanoic acid |
|---|
| InChI Identifier | InChI=1S/C34H38N4O4/c1-7-21-17(3)25-13-26-19(5)23(9-11-33(39)40)31(37-26)16-32-24(10-12-34(41)42)20(6)28(38-32)15-30-22(8-2)18(4)27(36-30)14-29(21)35-25/h13-16,35-36H,7-12H2,1-6H3,(H,39,40)(H,41,42)/b25-13-,26-13-,27-14-,28-15-,29-14-,30-15-,31-16-,32-16- |
|---|
| InChI Key | NCAJWYASAWUEBY-UJJXFSCMSA-N |
|---|
| Isomeric SMILES | CCC1=C(C)/C2=C/C3=C(CC)C(C)=C(N3)\C=C3/N=C(/C=C4\N=C(\C=C\1/N\2)C(C)=C4CCC(O)=O)C(CCC(O)=O)=C3C |
|---|
| Average Molecular Weight | 566.6899 |
|---|
| Monoisotopic Molecular Weight | 566.289305724 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as porphyrins. Porphyrins are compounds containing a fundamental skeleton of four pyrrole nuclei united through the alpha-positions by four methine groups to form a macrocyclic structure. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Tetrapyrroles and derivatives |
|---|
| Sub Class | Porphyrins |
|---|
| Direct Parent | Porphyrins |
|---|
| Alternative Parents | Not Available |
|---|
| Substituents | Not Available |
|---|
| Molecular Framework | Not Available |
|---|
| External Descriptors | Not Available |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 5 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| Not Available |