| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:33:10 UTC |
|---|
| Update date | 2017-01-19 02:36:39 UTC |
|---|
| FoodComEx ID | PC000873 |
|---|
| FoodDB Record | FDB002608 |
|---|
| Chemical Information |
|---|
| Name | Daidzein |
|---|
| Description | Widespread isoflavone in the Leguminosae, especies Phaseolus subspecies (broad beans, lima beans); also found in soy and soy products (tofu, miso), chick peas (Cicer arietinum) and peanuts (Arachis hypogaea). Nutriceutical with anticancer and bone protective props.
Daidzein is one of several known isoflavones. Isoflavones compounds are found in a number of plants, but soybeans and soy products like tofu and textured vegetable protein are the primary food source. Up until recently, daidzein was considered to be one of the most important and most studied isoflavones, however more recently attention has shifted to isoflavone metabolites. Equol represents the main active product of daidzein metabolism, produced via specific microflora in the gut. The clinical effectiveness of soy isoflavones may be a function of the ability to biotransform soy isoflavones to the more potent estrogenic metabolite, equol, which may enhance the actions of soy isoflavones, owing to its greater affinity for estrogen receptors, unique antiandrogenic properties, and superior antioxidant activity. However, not all individuals consuming daidzein produce equol. Only approximately one-third to one-half of the population is able to metabolize daidzein to equol. This high variability in equol production is presumably attributable to interindividual differences in the composition of the intestinal microflora, which may play an important role in the mechanisms of action of isoflavones. But, the specific bacterial species in the colon involved in the production of equol are yet to be discovered. (PMID: 18045128, 17579894, Curr Med Chem. 2007;14(26):2824-30., Mol Nutr Food Res. 2007 Jul;51(7):765-81.) Daidzein is a biomarker for the consumption of soy beans and other soy products.
. |
|---|
| CAS Number | 486-66-8 |
|---|
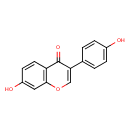
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| 4',7-Dihydroxy-isoflavone | HMDB | | 4',7-Dihydroxyisoflavone | db_source | | 7-Hydroxy-3-(4-hydroxy-phenyl)-chromone | biospider | | 7-Hydroxy-3-(4-hydroxyphenyl)-4-benzopyrone | biospider | | 7-Hydroxy-3-(4-hydroxyphenyl)-4H-1-benzopyran-4-one | ChEBI | | 7-Hydroxy-3-(4-hydroxyphenyl)-4H-1-benzopyran-4-one, 9CI | db_source | | 7-hydroxy-3-(4-hydroxyphenyl)-4H-chromen-4-one | biospider | | 7,4'-Dihydroxyisoflavone | biospider | | Daidzein | db_source | | Daidzeol | db_source | | Daizeol | db_source | | Dimethylbiochanin B | db_source | | Isoaurostatin | biospider | | K 251-6 | db_source | | Tatoin | db_source |
|
|---|
| Chemical Formula | C15H10O4 |
|---|
| IUPAC name | 7-hydroxy-3-(4-hydroxyphenyl)-4H-chromen-4-one |
|---|
| InChI Identifier | InChI=1S/C15H10O4/c16-10-3-1-9(2-4-10)13-8-19-14-7-11(17)5-6-12(14)15(13)18/h1-8,16-17H |
|---|
| InChI Key | ZQSIJRDFPHDXIC-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | OC1=CC=C(C=C1)C1=COC2=C(C=CC(O)=C2)C1=O |
|---|
| Average Molecular Weight | 254.241 |
|---|
| Monoisotopic Molecular Weight | 254.057908802 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as isoflavones. These are polycyclic compounds containing a 2-isoflavene skeleton which bears a ketone group at the C4 carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Isoflavonoids |
|---|
| Sub Class | Isoflav-2-enes |
|---|
| Direct Parent | Isoflavones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Isoflavone
- Hydroxyisoflavonoid
- Chromone
- Benzopyran
- 1-benzopyran
- Pyranone
- Phenol
- 1-hydroxy-2-unsubstituted benzenoid
- Benzenoid
- Monocyclic benzene moiety
- Pyran
- Heteroaromatic compound
- Oxacycle
- Organoheterocyclic compound
- Organooxygen compound
- Organic oxygen compound
- Hydrocarbon derivative
- Organic oxide
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Mp 330° (323°) | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 50 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | C449 |
|---|
| AKSci | J10637 |
|---|
| AKSci | HMDB0003312 |
|---|
| Cayman Chemical | 10005166 |
|---|
| MetaSci | HMDB0003312 |
|---|
| Toronto Research Chemicals | D103500 |
|---|