| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:33:06 UTC |
|---|
| Update date | 2017-01-19 02:36:39 UTC |
|---|
| FoodComEx ID | PC000853 |
|---|
| FoodDB Record | FDB023181 |
|---|
| Chemical Information |
|---|
| Name | 3,5-Diiodo-L-tyrosine |
|---|
| Description | A product from the iodination of monoiodotyrosine. In the biosynthesis of thyroid hormones, diiodotyrosine residues are coupled with other monoiodotyrosine or diiodotyrosine residues to form T4 or T3 thyroid hormones (thyroxine and triiodothyronine). [HMDB] |
|---|
| CAS Number | 300-39-0 |
|---|
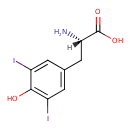
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (2S)-2-amino-3-(4-Hydroxy-3,5-diiodophenyl)propanoate | Generator | | (2S)-2-amino-3-(4-Hydroxy-3,5-diiodophenyl)propanoic acid | ChEBI | | 3,5-Diiodotyrocine | hmdb | | 3,5-Diiodotyrosine | ChEBI | | 3,5-Iodo-L-tyrosine | hmdb | | 3,5-L-Diiodotyrosine | hmdb | | 4-Hydroxy-3,5-diiodophenylalanine | hmdb | | Diiodotyrosine | ChEBI | | DiIY | ChEBI | | DIT | ChEBI | | L-3,5-DiioDOTyrosine | hmdb | | L-Diiodotyrosine | hmdb |
|
|---|
| Chemical Formula | C9H9I2NO3 |
|---|
| IUPAC name | (2S)-2-amino-3-(4-hydroxy-3,5-diiodophenyl)propanoic acid |
|---|
| InChI Identifier | InChI=1S/C9H9I2NO3/c10-5-1-4(2-6(11)8(5)13)3-7(12)9(14)15/h1-2,7,13H,3,12H2,(H,14,15)/t7-/m0/s1 |
|---|
| InChI Key | NYPYHUZRZVSYKL-ZETCQYMHSA-N |
|---|
| Isomeric SMILES | [H][C@](N)(CC1=CC(I)=C(O)C(I)=C1)C(O)=O |
|---|
| Average Molecular Weight | 432.9816 |
|---|
| Monoisotopic Molecular Weight | 432.867179999 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as tyrosine and derivatives. Tyrosine and derivatives are compounds containing tyrosine or a derivative thereof resulting from reaction of tyrosine at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Tyrosine and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Tyrosine or derivatives
- Phenylalanine or derivatives
- 3-phenylpropanoic-acid
- Amphetamine or derivatives
- Alpha-amino acid
- L-alpha-amino acid
- 2-iodophenol
- 2-halophenol
- Aralkylamine
- Halobenzene
- Phenol
- Iodobenzene
- Aryl iodide
- Monocyclic benzene moiety
- Benzenoid
- Aryl halide
- Amino acid
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Amine
- Primary aliphatic amine
- Organonitrogen compound
- Organooxygen compound
- Primary amine
- Organic nitrogen compound
- Carbonyl group
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Organoiodide
- Organohalogen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 2 g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | 8172AF |
|---|
| AKSci | F525 |
|---|
| AKSci | J10902 |
|---|
| AKSci | J40127 |
|---|
| AKSci | J93316 |
|---|
| Toronto Research Chemicals | D466055 |
|---|