| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:32:53 UTC |
|---|
| Update date | 2017-01-19 02:36:37 UTC |
|---|
| FoodComEx ID | PC000814 |
|---|
| FoodDB Record | FDB001114 |
|---|
| Chemical Information |
|---|
| Name | alpha,alpha-Trehalose |
|---|
| Description | Occurs in fungi. EU and USA approved sweetener
Trehalose is a disaccharide formed by a 1, 1-glucoside bond between two ?-glucose units. Because trehalose is formed by the bonding of two reducing groups, it has no capacity to reduce other compounds.; Trehalose was first isolated from ergot of rye. Emil Fischer first described the trehalose-hydrolyzing enzyme in yeast. Trehalose is a non-reducing sugar formed from two glucose units joined by a 1-1 alpha bond giving it the name of ?-D-glucopyranosyl-(1?1)-?-D-glucopyranoside. The bonding makes trehalose very resistant to acid hydrolysis, and therefore stable in solution at high temperatures even under acidic conditions. The bonding also keeps non-reducing sugars in closed-ring form, such that the aldehyde or ketone end-groups do not bind to the lysine or arginine residues of proteins (a process called glycation). Trehalose is broken down by the enzyme trehalase into glucose. Trehalose has about 45% the sweetness of sucrose. Trehalose is less soluble than sucrose, except at high temperatures (>80 °C). Trehalose forms a rhomboid crystal as the dihydrate, and has 90% of the calorific content of sucrose in that form. Anhydrous forms of trehalose readily regain moisture to form the dihydrate. Anhydrous forms of trehalose can show interesting physical properties when heat-treated.; Trehalose, also known as mycose, is a 1-alpha (disaccharide) sugar found extensively but not abundantly in nature. It is thought to be implicated in anhydrobiosis - the ability of plants and animals to withstand prolonged periods of desiccation. The sugar is thought to form a gel phase as cells dehydrate, which prevents disruption of internal cell organelles by effectively splinting them in position. Rehydration then allows normal cellular activity to be resumed without the major, generally lethal damage that would normally follow a dehydration/reyhdration cycle.; Trehalose is a non-reducing sugar formed from two glucose units joined by a 1-1 alpha bond giving it the name of alpha-D-glucopyranoglucopyranosyl-1,1-alpha-D-glucopyranoside. The bonding makes trehalose very resistant to acid hydrolysis, and therefore stable in solution at high temperatures even under acidic conditions. The bonding also keeps non-reducing sugars in closed-ring form, such that the aldehyde or ketone end-groups do not bind to the lysine or arginine residues of proteins (a process called glycation). The enzyme trehalase, present but not abundant in most people, breaks it into two glucose molecules, which can then be readily absorbed in the gut.; Trehalose is an important components of insects circulating fluid.It acts as a storage form of insect circulating fluid and it is important in respiration.; Trehalose, also known as mycose, is a natural alpha-linked disaccharide formed by an ?, ?-1, 1-glucoside bond between two ?-glucose units. In 1832 Wiggers discovered trehalose in an ergot of rye and in 1859 Berthelot isolated it from trehala manna, a substance made by weevils, and named it trehalose. It can be synthesised by fungi, plants, and invertebrate animals. It is implicated in anhydrobiosis ? the ability of plants and animals to withstand prolonged periods of desiccation. It has high water retention capabilities and is used in food and cosmetics. The sugar is thought to form a gel phase as cells dehydrate, which prevents disruption of internal cell organelles by effectively splinting them in position. Rehydration then allows normal cellular activity to be resumed without the major, lethal damage that would normally follow a dehydration/re-hydration cycle. Trehalose has the added advantage of being an antioxidant. Extracting trehalose used to be a difficult and costly process, but, recently, the Hayashibara company (Okayama, Japan) confirmed an inexpensive extraction technology from starch for mass production. Trehalose is currently being used for a broad spectrum of applications. alpha,alpha-Trehalose is found in bitter gourd, mushrooms, and garden tomato. |
|---|
| CAS Number | 99-20-7 |
|---|
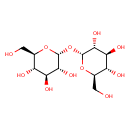
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (Glc)2 | ChEBI | | α-d-glucopyranoside, α-d-glucopyranosyl | biospider | | α-d-glucopyranosyl-α-d-glucopyranoside | biospider | | α-d-trehalose | biospider | | α-trehalose | biospider | | α,α-trehalose | biospider | | α,α'-trehalose | biospider | | a-D-Glcp-(11)-a-D-glcp | Generator | | a-D-Glucopyranosyl-a-D-glucopyranoside | Generator | | a-D-Glucopyranosyl-a-D-glucopyranoside, 9CI, 8CI | db_source | | a-D-Trehalose | Generator | | a-Trehalose | Generator | | a,a-Trehalose | Generator | | a,Alpha'-trehalose | Generator | | alpha-D-Glcp-(11)-alpha-D-glcp | ChEBI | | Alpha-d-glucopyranosyl beta-d-glucopyranoside | biospider | | alpha-D-Glucopyranosyl-alpha-D-glucopyranoside | ChEBI | | alpha-D-Trehalose | ChEBI | | alpha-Trehalose | ChEBI | | Alpha,alpha-trehalose | biospider | | Alpha,alpha'-trehalose | biospider | | Alpha,beta-trehalose | biospider | | D-(+)-trehalose | biospider | | D-trehalose-anhydrous | biospider | | Delta-trehalose-anhydrous | biospider | | Ergot sugar | biospider | | Hexopyranosyl hexopyranoside | biospider | | Mushroom sugar | db_source | | Mycose | db_source | | Natural trehalose | biospider | | Trehalose | db_source | | Trehalose, dihydrate | biospider | | α-D-glcp-(11)-α-D-glcp | Generator | | α-D-glucopyranosyl-α-D-glucopyranoside | Generator | | α-D-trehalose | Generator | | α-trehalose | Generator | | α,alpha'-trehalose | Generator | | α,α-trehalose | Generator |
|
|---|
| Chemical Formula | C12H22O11 |
|---|
| IUPAC name | (2R,3S,4S,5R,6R)-2-(hydroxymethyl)-6-{[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxane-3,4,5-triol |
|---|
| InChI Identifier | InChI=1S/C12H22O11/c13-1-3-5(15)7(17)9(19)11(21-3)23-12-10(20)8(18)6(16)4(2-14)22-12/h3-20H,1-2H2 |
|---|
| InChI Key | HDTRYLNUVZCQOY-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | OCC1OC(OC2OC(CO)C(O)C(O)C2O)C(O)C(O)C1O |
|---|
| Average Molecular Weight | 342.2965 |
|---|
| Monoisotopic Molecular Weight | 342.116211546 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as o-glycosyl compounds. These are glycoside in which a sugar group is bonded through one carbon to another group via a O-glycosidic bond. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | O-glycosyl compounds |
|---|
| Alternative Parents | |
|---|
| Substituents | - O-glycosyl compound
- Disaccharide
- Oxane
- Secondary alcohol
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Acetal
- Hydrocarbon derivative
- Primary alcohol
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Mp 214-216° (anhyd.) | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 2 g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | liquid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | 6953AC |
|---|