| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:32:29 UTC |
|---|
| Update date | 2017-01-19 02:36:35 UTC |
|---|
| FoodComEx ID | PC000751 |
|---|
| FoodDB Record | FDB011904 |
|---|
| Chemical Information |
|---|
| Name | Quercetin |
|---|
| Description | Quercetin, also known as sophoretin or xanthaurine, belongs to the class of organic compounds known as flavonols. Flavonols are compounds that contain a flavone (2-phenyl-1-benzopyran-4-one) backbone carrying a hydroxyl group at the 3-position. Quercetin is an antioxidant, like many other phenolic heterocyclic compounds. Quercetin is a very hydrophobic molecule, practically insoluble in water, and relatively neutral. Quercetin is a widely distributed flavonoid found in many plants and fruits including red grapes, citrus fruit, tomatoes, broccoli, red onions, kale, various leafy green vegetables, and a number of berries, including raspberries and cranberries. In plants quercetin functions as a naturally occurring polar auxin transport inhibitor (PMID: 12237347). Quercetin has a bitter flavor and is used as an ingredient in dietary supplements, beverages, and foods. Quercetin itself (the aglycone), as opposed to quercetin glycosides, is not a normal dietary component. Quercitin glycosides are converted to phenolic acids as they pass through the gastrointestinal tract. In the human body quercetin functions as a non-specific protein kinase enzyme inhibitor (PMID: 15019969). It also acts as a phytoestrogen and has been reported to have estrogenic activities by activating both estrogen receptor alpha (ER alpha) and estrogen beta (ER beta) (PMID: 17724002). In human breast cancer cell lines, quercetin has been found to act as an agonist of the G protein-coupled estrogen receptor (GPER) (PMID: 15090535). Despite these many known interactions with human proteins, quercetin has not been confirmed scientifically as a specific therapeutic for any condition nor been approved by any regulatory agency. In particular, the U.S. Food and Drug Administration has not approved any health claims for quercetin. Nevertheless, there is a clear inverse correlation between dietary consumption of flavonols and flavones and reduced incidence and mortality from cardiovascular disease and cancer. In recent years, a large amount of experimental and some clinical data have accumulated regarding the effects of flavonoids on the endothelium under physiological and pathological conditions. The meta-analysis of seven prospective cohort studies concluded that the individuals in the top third of dietary flavonol intake are associated with a reduced risk of mortality from coronary heart disease as compared with those in the bottom third, after adjustment for known risk factors and other dietary components. A limited number of intervention studies with flavonoids and flavonoid containing foods and extracts has been performed in several pathological conditions. (PMID: 17015250). |
|---|
| CAS Number | 117-39-5 |
|---|
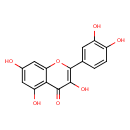
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| 2-(3,4-Dihydroxy-phenyl)-3,5,7-trihydroxy-chromen-4-one | HMDB | | 2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-4H-1-benzopyran-4-one | ChEBI | | 2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-4H-1-benzopyran-4-one, 9CI | db_source | | 2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxy-4H-chromen-4-one | biospider | | 2-(3,4-Dihydroxyphenyl)-3,5,7-trihydroxychromen-4-one | biospider | | 3,3',4,5,7-Pentahydroxyflavone | biospider | | 3,3',4',5,7-Pentahydroxyflavone | biospider | | 3,4',5,5',7-Pentahydroxy-flavone | HMDB | | 3,5,7-Trihydroxy-2-(3,4-dihydroxyphenyl)-4H-chromen-4-on | HMDB | | 3,5,7-Trihydroxy-2-(3,4-dihydroxyphenyl)-4H-chromen-4-one | biospider | | 3,5,7,3',4'-Pentahydroxyflavone | biospider | | 3',4',5,7-Tetrahydroxyflavan-3-ol | HMDB | | 3',4',5,7-Tetrahydroxyflavon-3-ol | biospider | | 3',4',5,7-Tetrahydroxyflavonol | db_source | | Flavin meletin | HMDB | | Flavone, 3,4',5,5',7-pentahydroxy- | biospider | | Meletin | db_source | | Quercetin | db_source | | Quercetin dihydrate | HMDB | | Quercetine | biospider | | Quercetol | db_source | | Quercitin | biospider | | Quertin | db_source | | Quertine | biospider | | Sophoretin | db_source | | Xanthaurine | biospider |
|
|---|
| Chemical Formula | C15H10O7 |
|---|
| IUPAC name | 2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4H-chromen-4-one |
|---|
| InChI Identifier | InChI=1S/C15H10O7/c16-7-4-10(19)12-11(5-7)22-15(14(21)13(12)20)6-1-2-8(17)9(18)3-6/h1-5,16-19,21H |
|---|
| InChI Key | REFJWTPEDVJJIY-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | OC1=CC2=C(C(O)=C1)C(=O)C(O)=C(O2)C1=CC=C(O)C(O)=C1 |
|---|
| Average Molecular Weight | 302.2357 |
|---|
| Monoisotopic Molecular Weight | 302.042652674 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as flavonols. Flavonols are compounds that contain a flavone (2-phenyl-1-benzopyran-4-one) backbone carrying a hydroxyl group at the 3-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | Flavones |
|---|
| Direct Parent | Flavonols |
|---|
| Alternative Parents | |
|---|
| Substituents | - 3-hydroxyflavone
- 3'-hydroxyflavonoid
- 3-hydroxyflavonoid
- 4'-hydroxyflavonoid
- 5-hydroxyflavonoid
- 7-hydroxyflavonoid
- Hydroxyflavonoid
- Chromone
- Benzopyran
- 1-benzopyran
- Catechol
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Pyranone
- Benzenoid
- Monocyclic benzene moiety
- Pyran
- Heteroaromatic compound
- Vinylogous acid
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Organic oxide
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | 0.06 mg/mL at 16 oC | SEIDELL,A (1941) |
|---|
| Melting Point | Mp 313-314° dec. | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 2 . |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | S295 |
|---|
| Cayman Chemical | 10005169 |
|---|
| Cayman Chemical | HMDB0005794 |
|---|
| Glentham | GP9232 |
|---|
| MetaSci | HMDB0005794 |
|---|
| Toronto Research Chemicals | Q510038 |
|---|