| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:32:20 UTC |
|---|
| Update date | 2017-01-19 02:36:34 UTC |
|---|
| FoodComEx ID | PC000738 |
|---|
| FoodDB Record | FDB014297 |
|---|
| Chemical Information |
|---|
| Name | (R)-Limonene |
|---|
| Description | Major constituent of oil of orange rind, dill oil, oil of cumin, bergamot, caraway, lemon balm and lemon. obtained comly. from orange oil on a large scale (ca. 50000 T/a). Flavouring agent. Nutriceutical with anticancer props. (R)-Limonene is found in many foods, some of which are citrus, nutmeg, guava, and cardamom. |
|---|
| CAS Number | 5989-27-5 |
|---|
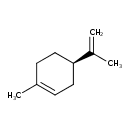
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (+)-(4R)-Limonene | biospider | | (+)-(r)-limonene | biospider | | (+)-1,8-para-Menthadiene | biospider | | (+)-4-Isopropenyl-1-methylcyclohexene | biospider | | (+)-carvene | biospider | | (+)-limonene | biospider | | (+)-p-Mentha-1,8-diene | biospider | | (+)-r-limonene | biospider | | (4R)-(+)-Limonene | biospider | | (4R)-1-methyl-4-(1-methylethenyl)cyclohexene | biospider | | (4R)-1-methyl-4-(prop-1-en-2-yl)cyclohex-1-ene | biospider | | (4R)-1-methyl-4-isopropenylcyclohex-1-ene | biospider | | (4R)-4-isopropenyl-1-methylcyclohexene | biospider | | (4R)-Limonene | biospider | | (d)-limonene | biospider | | (r)-(+)-limonene | biospider | | (R)-(+)-p-Mentha-1,8-diene | biospider | | (R)-1-methyl-4-(1-methylethenyl)cyclohexene | biospider | | (R)-4-Isopropenyl-1-methyl-1-cyclohexene | biospider | | (r)-limonene | biospider | | (R)-p-Mentha-1,8-diene | biospider | | 1-Methyl-4-(1-methylethenyl)cyclohexene, (R)- | biospider | | 4betaH-p-mentha-1,8-diene | manual | | Cyclohexene, 1-methyl-4-(1-methylethenyl)-, (4R)- | biospider | | Cyclohexene, 1-methyl-4-(1-methylethenyl)-, (R)- | biospider | | D-(+)-limonene | biospider | | D-1,8-p-Menthadiene | biospider | | D-limonen | biospider | | D-limonene | biospider | | d-p-Mentha-1,8-diene | biospider | | Dextro-limonene | biospider | | p-Mentha-1,8-diene, (R)-(+)- | biospider |
|
|---|
| Chemical Formula | C10H16 |
|---|
| IUPAC name | (4R)-1-methyl-4-(prop-1-en-2-yl)cyclohex-1-ene |
|---|
| InChI Identifier | InChI=1S/C10H16/c1-8(2)10-6-4-9(3)5-7-10/h4,10H,1,5-7H2,2-3H3/t10-/m0/s1 |
|---|
| InChI Key | XMGQYMWWDOXHJM-JTQLQIEISA-N |
|---|
| Isomeric SMILES | CC(=C)[C@@H]1CCC(C)=CC1 |
|---|
| Average Molecular Weight | 136.234 |
|---|
| Monoisotopic Molecular Weight | 136.125200512 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as menthane monoterpenoids. These are monoterpenoids with a structure based on the o-, m-, or p-menthane backbone. P-menthane consists of the cyclohexane ring with a methyl group and a (2-methyl)-propyl group at the 1 and 4 ring position, respectively. The o- and m- menthanes are much rarer, and presumably arise by alkyl migration of p-menthanes. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Monoterpenoids |
|---|
| Direct Parent | Menthane monoterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - P-menthane monoterpenoid
- Monocyclic monoterpenoid
- Branched unsaturated hydrocarbon
- Cycloalkene
- Cyclic olefin
- Unsaturated aliphatic hydrocarbon
- Unsaturated hydrocarbon
- Olefin
- Hydrocarbon
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework | Aliphatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | 4.57 | LI,J & PURDUE,EM (1995) |
|---|
| Experimental Water Solubility | 0.0138 mg/mL at 25 oC | MASSALDI,HA & KING,CJ (1973) |
|---|
| Melting Point | -74.3 oC | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 30 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | liquid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | 6458AE |
|---|
| MetaSci | HMDB0004321 |
|---|
| Sigma-Aldrich | HMDB0004321 |
|---|
| Toronto Research Chemicals | L461740 |
|---|