| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:32:10 UTC |
|---|
| Update date | 2017-01-19 02:36:33 UTC |
|---|
| FoodComEx ID | PC000714 |
|---|
| FoodDB Record | FDB014499 |
|---|
| Chemical Information |
|---|
| Name | (-)-Menthol |
|---|
| Description | Present in large amts. in peppermint oil (Mentha piperita), also in other Mentha subspecies. It is used in confectionery and perfumery. Flavouring agent
(-)-menthol is the major occuring menthol stereoisomer in nature. (-)-Menthol is found in many foods, some of which are lovage, sweet basil, sweet marjoram, and orange mint. |
|---|
| CAS Number | 2216-51-5 |
|---|
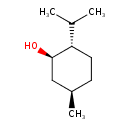
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (-)-(1R,3R,4S)-Menthol | biospider | | (-)-Menthol | db_source | | (-)-menthyl alcohol | biospider | | (-)-trans-p-menthan-cis-ol | biospider | | (1a,2b,5a)-5-Methyl-2(1-methylethyl)cyclohexanol | Generator | | (1alpha,2beta,5alpha)-5-methyl-2(1-methylethyl)cyclohexanol | biospider | | (1R-(1-a,2-b,5-a))-5-Methyl-2-(1-methylethyl)cyclohexanol | Generator | | (1R-(1-alpha,2-beta,5-alpha))-5-Methyl-2-(1-methylethyl)cyclohexanol | ChEBI | | (1R-(1-α,2-β,5-α))-5-methyl-2-(1-methylethyl)cyclohexanol | Generator | | (1R,2S,5R)-(-)-Menthol | HMDB | | (1R,2S,5R)-2-isopropyl-5-methylcyclohexanol | biospider | | (1R,2S,5R)-5-methyl-2-(1-methylethyl)cyclohexanol | biospider | | (1R,2S,5R)-5-methyl-2-(propan-2-yl)cyclohexan-1-ol | biospider | | (1R,2S,5R)-5-methyl-2-propan-2-yl-cyclohexan-1-ol | biospider | | (1R,2S,5R)-5-methyl-2-propan-2-ylcyclohexan-1-ol | biospider | | (1R,3R,4S)-(-)-MENTHOL | biospider | | (1R)-(-)-Menthol | biospider | | (1α,2β,5α)-5-methyl-2(1-methylethyl)cyclohexanol | Generator | | (R)-(-)-Menthol | HMDB | | 1-Menthol | HMDB | | 5-Methyl-2-propan-2-ylcyclohexan-1-ol | HMDB | | Cyclohexanol, 5-methyl-2-(1-methylethyl)-, (1R,2S,5R)- | biospider | | Cyclohexanol, 5-methyl-2-(1-methylethyl)-, (1R,3R,4S)- | biospider | | D-(-)-Menthol | HMDB | | FEMA 2665 | db_source | | Hexahydrothymol | HMDB | | L-(-)-menthol | biospider | | L-menthol | biospider | | Levomenthol | biospider | | Levomentholum | ChEBI | | Levomentol | ChEBI | | Menthacamphor | db_source | | Menthol, (1R,3R,4S)-(-)- | biospider | | Menthol, l- | biospider | | Menthomenthol | HMDB | | P-Menthan-3-ol | HMDB | | Peppermint camphor | db_source | | U.S.p. menthol | HMDB |
|
|---|
| Chemical Formula | C10H20O |
|---|
| IUPAC name | (1R,2S,5R)-5-methyl-2-(propan-2-yl)cyclohexan-1-ol |
|---|
| InChI Identifier | InChI=1S/C10H20O/c1-7(2)9-5-4-8(3)6-10(9)11/h7-11H,4-6H2,1-3H3/t8-,9+,10-/m1/s1 |
|---|
| InChI Key | NOOLISFMXDJSKH-KXUCPTDWSA-N |
|---|
| Isomeric SMILES | CC(C)[C@@H]1CC[C@@H](C)C[C@H]1O |
|---|
| Average Molecular Weight | 156.269 |
|---|
| Monoisotopic Molecular Weight | 156.151415264 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as menthane monoterpenoids. These are monoterpenoids with a structure based on the o-, m-, or p-menthane backbone. P-menthane consists of the cyclohexane ring with a methyl group and a (2-methyl)-propyl group at the 1 and 4 ring position, respectively. The o- and m- menthanes are much rarer, and presumably arise by alkyl migration of p-menthanes. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Monoterpenoids |
|---|
| Direct Parent | Menthane monoterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - P-menthane monoterpenoid
- Monocyclic monoterpenoid
- Cyclohexanol
- Cyclic alcohol
- Secondary alcohol
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Alcohol
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework | Aliphatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | 3.40 | GRIFFIN,S ET AL. (1999) |
|---|
| Experimental Water Solubility | 0.49 mg/mL at 25 oC | CHEMICALS INSPECTION AND TESTING INSTITU (1992) |
|---|
| Melting Point | Mp 42.5-43° | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 500 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | liquid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | X7298 |
|---|
| Glentham | GK4093 |
|---|
| Toronto Research Chemicals | M218875 |
|---|