| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:32:01 UTC |
|---|
| Update date | 2017-01-19 02:36:33 UTC |
|---|
| FoodComEx ID | PC000696 |
|---|
| FoodDB Record | FDB003357 |
|---|
| Chemical Information |
|---|
| Name | Oxalic acid |
|---|
| Description | Present in many plants and vegetables. Toxin from rhubarb, especies the leaves but also present in the stems
A strong dicarboxylic acid occurring in many plants and vegetables. It is produced in the body by metabolism of glyoxylic acid or ascorbic acid. It is not metabolized but excreted in the urine. It is used as an analytical reagent and general reducing agent. -- Pubchem; Bodily oxalic acid may also be synthesized via the metabolism of either glyoxylic acid or unused ascorbic acid (vitamin C), which is a serious health consideration for long term megadosers of vitamin C supplements. 80% of kidney stones are formed from calcium oxalate. Some Aspergillus species produce oxalic acid, which reacts with blood or tissue calcium to precipitate calcium oxalate. There is some preliminary evidence that the administration of probiotics can affect oxalic acid excretion rates (and presumably oxalic acid levels as well.) -- Wikipedia; Oxalate, the conjugate base of oxalic acid, is an excellent ligand for metal ions. It usually binds as a bidentate ligand forming a 5-membered MO2C2 ring. An illustrative complex is potassium ferrioxalate, K3[Fe(C2O4)3]. The drug Oxaliplatin exhibits improved water solubility relative to older platinum-based drugs, avoiding the dose-limiting side-effect of nephrotoxicity. Oxalic acid and oxalates can be oxidized by permanganate in an autocatalytic reaction.; Oxalic acid (IUPAC name: ethanedioic acid, formula H2C2O4) is a dicarboxylic acid with structure (HOOC)-(COOH). Because of the joining of two carboxyl groups, this is one of the strongest organic acids. It is also a reducing agent. The anions of oxalic acid as well as its salts and esters are known as oxalates. -- Wikipedia; Oxalic acid is an important reagent in lanthanide chemistry. Hydrated lanthanide oxalates form readily in strongly acid solution in a densely crystalline easily filtered form, largely free from contamination by non-lanthanide elements. Lanthanide oxalates figure importantly in commercial processing of lanthanides, and are used to recover lanthanides from solution after separation. Upon ignition, lanthanide oxalates are converted to the oxides, which are the most common form in which the lanthanides are marketed.; Oxalic acid is the chemical compound with the formula H2C2O4. This dicarboxylic acid is better described with the formula HOOCCOOH. It is a relatively strong organic acid, being about 3,000 times as strong as acetic acid. The di-anion, known as oxalate, is also a reducing agent as well as a ligand in coordination chemistry. Many metal ions form insoluble precipitates with oxalate, a prominent example being calcium oxalate, which is the primary constituent of the most common kind of kidney stones. Oxalic acid is found in many foods, some of which are lettuce, chinese chives, red beetroot, and cherry tomato. |
|---|
| CAS Number | 144-62-7 |
|---|
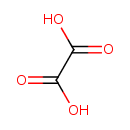
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| 2dua | biospider | | 2hwg | biospider | | 553-91-3 (DILITHIUM) | biospider | | Acido ossalico | biospider | | Acidum oxalicum | biospider | | Acidum Oxalicum-Injeel Forte Liq (D6-D200) | biospider | | Aktisal | biospider | | Ammonium oxalate;Ethane-1,2-dioate | biospider | | Aquisal | biospider | | Clover acid | db_source | | Ethandisaeure | ChEBI | | Ethane-1,2-dioate | Generator | | Ethane-1,2-dioic acid | biospider | | Ethanedioate | biospider | | Ethanedioic acid | ChEBI | | Ethanedioic acid (9CI) | biospider | | Ethanedioic acid dihydrate | biospider | | Ethanedioic acid, 9CI | db_source | | Ethanedionate | biospider | | Ethanedionic acid | biospider | | H2ox | biospider | | Hooccooh | biospider | | Kyselina stavelova | HMDB | | Magnesium permanganate | biospider | | Oxaalzuur | biospider | | Oxagel | biospider | | Oxalate | Generator | | Oxalate standard for ic | biospider | | Oxalic acid (8CI) | biospider | | Oxalic acid 2-Hydrate | biospider | | Oxalic acid anhydrous | biospider | | Oxalic acid diammonium salt | biospider | | Oxalic acid dihydrate | biospider | | Oxalic Acid, Cadmium Salt (1:1) | biospider | | Oxalicum 5dh-30ch | biospider | | Oxalicum acidum | biospider | | Oxalsaeure | ChEBI | | OXD | biospider | | Oxiric acid | biospider | | OXL | biospider | | Ultraplast Activate S 52 | biospider |
|
|---|
| Chemical Formula | C2H2O4 |
|---|
| IUPAC name | oxalic acid |
|---|
| InChI Identifier | InChI=1S/C2H2O4/c3-1(4)2(5)6/h(H,3,4)(H,5,6) |
|---|
| InChI Key | MUBZPKHOEPUJKR-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | OC(=O)C(O)=O |
|---|
| Average Molecular Weight | 90.0349 |
|---|
| Monoisotopic Molecular Weight | 89.995308552 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as dicarboxylic acids and derivatives. These are organic compounds containing exactly two carboxylic acid groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Dicarboxylic acids and derivatives |
|---|
| Direct Parent | Dicarboxylic acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Dicarboxylic acid or derivatives
- Carboxylic acid
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | 220 mg/mL at 25 oC | YALKOWSKY,SH & DANNENFELSER,RM (1992) |
|---|
| Melting Point | Mp 101.5° (dihydrate) | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 1 . |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | H158 |
|---|
| Glentham | GK3343 |
|---|
| MetaSci | HMDB0002329 |
|---|
| Sigma-Aldrich | HMDB0002329 |
|---|
| Toronto Research Chemicals | O845120 |
|---|