| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:31:19 UTC |
|---|
| Update date | 2017-01-19 02:36:28 UTC |
|---|
| FoodComEx ID | PC000561 |
|---|
| FoodDB Record | FDB022747 |
|---|
| Chemical Information |
|---|
| Name | Ranitidine |
|---|
| Description | A non-imidazole blocker of those histamine receptors that mediate gastric secretion (H2 receptors). It is used to treat gastrointestinal ulcers. -- Pubchem; Ranitidine is a histamine H2-receptor antagonist that inhibits stomach acid production, and commonly used in the treatment of peptic ulcer disease (PUD) and gastroesophageal reflux disease (GERD). It is currently marketed by GlaxoSmithKline under the trade name Zantac. -- Wikipedia [HMDB] |
|---|
| CAS Number | 66357-59-3 |
|---|
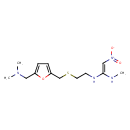
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| Achedos | hmdb | | acidex | hmdb | | Atural | hmdb | | Axoban | hmdb | | Coralen | hmdb | | Curan | hmdb | | Duractin | hmdb | | Ezopta | hmdb | | Gastrial | hmdb | | Gastrosedol | hmdb | | Istomar | hmdb | | Logast | hmdb | | Mauran | hmdb | | Microtid | hmdb | | Ptinolin | hmdb | | Quantor | hmdb | | Quicran | hmdb | | Radinat | hmdb | | Randin | hmdb | | Ranidine | hmdb | | Ranin | hmdb | | Raniogas | hmdb | | Ranisen | hmdb | | Raniter | hmdb | | Ranitidina | ChEBI | | Ranitidina [INN-Spanish] | hmdb | | Ranitidine | hmdb | | Ranitidine (Form I And Form Ii) | hmdb | | Ranitidine [USAN:BAN:INN] | hmdb | | Ranitidine Base | hmdb | | Ranitidine HCL 1/2 Type | hmdb | | Ranitidinum | ChEBI | | Ranitidinum [INN-Latin] | hmdb | | Ranitiget | hmdb | | Rantacid | hmdb | | Rantidine | hmdb | | Ratic | hmdb | | Raticina | hmdb | | RND | hmdb | | Sampep | hmdb | | Taural | hmdb | | Ul-Pep | hmdb | | Ulceranin | hmdb | | Urantac | hmdb | | Verlost | hmdb | | Vesyca | hmdb | | Vizerul | hmdb | | Weichilin | hmdb | | Weidos | hmdb | | Xanidine | hmdb | | Zantab | hmdb | | ZANTAC | hmdb | | Zantac (TN) | hmdb | | Zantadin | hmdb |
|
|---|
| Chemical Formula | C13H22N4O3S |
|---|
| IUPAC name | dimethyl[(5-{[(2-{[(E)-1-(methylamino)-2-nitroethenyl]amino}ethyl)sulfanyl]methyl}furan-2-yl)methyl]amine |
|---|
| InChI Identifier | InChI=1S/C13H22N4O3S/c1-14-13(9-17(18)19)15-6-7-21-10-12-5-4-11(20-12)8-16(2)3/h4-5,9,14-15H,6-8,10H2,1-3H3/b13-9+ |
|---|
| InChI Key | VMXUWOKSQNHOCA-UKTHLTGXSA-N |
|---|
| Isomeric SMILES | CN\C(NCCSCC1=CC=C(CN(C)C)O1)=C/[N+]([O-])=O |
|---|
| Average Molecular Weight | 314.404 |
|---|
| Monoisotopic Molecular Weight | 314.14126128 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as aralkylamines. These are alkylamines in which the alkyl group is substituted at one carbon atom by an aromatic hydrocarbyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic nitrogen compounds |
|---|
| Class | Organonitrogen compounds |
|---|
| Sub Class | Amines |
|---|
| Direct Parent | Aralkylamines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aralkylamine
- Furan
- Heteroaromatic compound
- C-nitro compound
- Tertiary amine
- Tertiary aliphatic amine
- Organic nitro compound
- Secondary aliphatic amine
- Organic oxoazanium
- Secondary amine
- Thioether
- Sulfenyl compound
- Allyl-type 1,3-dipolar organic compound
- Propargyl-type 1,3-dipolar organic compound
- Organic 1,3-dipolar compound
- Oxacycle
- Organoheterocyclic compound
- Dialkylthioether
- Organic zwitterion
- Organooxygen compound
- Organosulfur compound
- Organic oxygen compound
- Organic oxide
- Organopnictogen compound
- Hydrocarbon derivative
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 200 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| Not Available |