| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:31:14 UTC |
|---|
| Update date | 2017-01-19 02:36:27 UTC |
|---|
| FoodComEx ID | PC000535 |
|---|
| FoodDB Record | FDB022728 |
|---|
| Chemical Information |
|---|
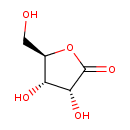
| Name | Ribonolactone |
|---|
| Description | Ribonolactone is a metabolite normally not detectable in human biofluids; however, it has been found in the urine of patients with neuroblastoma. (PMID 699273) [HMDB] |
|---|
| CAS Number | 5336-08-3 |
|---|
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| D-(+)-Ribonate g-lactone | Generator | | D-(+)-Ribonate gamma-lactone | Generator | | D-(+)-Ribonate γ-lactone | Generator | | D-(+)-Ribonic acid g-Lactone | hmdb | | D-(+)-Ribonic acid gamma-lactone | hmdb | | D-(+)-Ribonic acid γ-lactone | Generator | | D-(+)-Ribonone-1.4-lactone | hmdb | | D-Ribonate-1,4-lactone | Generator | | D-Ribonic acid-1,4-lactone | ChEBI | | D-Ribono-1,4-lactone | hmdb | | D-Ribono-g-lactone | Generator | | D-Ribono-gamma-lactone | hmdb | | D-Ribono-γ-lactone | Generator | | D-Ribonolactone | hmdb | | D-Ribopentono-1,4-lactone | hmdb | | D(+)-Ribonic acid gamma-lactone | hmdb | | delta-(+)-Ribonic acid g-Lactone | hmdb | | delta-(+)-Ribonic acid gamma-lactone | hmdb | | delta-(+)-Ribonone-1.4-lactone | hmdb | | delta-Ribono-1,4-lactone | hmdb | | delta-Ribono-gamma-lactone | hmdb | | delta-Ribonolactone | hmdb | | delta-Ribopentono-1,4-lactone | hmdb | | Deoxyribonolactone | hmdb | | gamma-Lactone of ribonate | hmdb | | gamma-Lactone of ribonic acid | hmdb | | gamma-lactone-Ribonate | hmdb | | gamma-lactone-Ribonic acid | hmdb | | Ribono-g-lactone | Generator | | Ribono-gamma-lactone | hmdb | | Ribono-γ-lactone | Generator |
|
|---|
| Chemical Formula | C5H8O5 |
|---|
| IUPAC name | (3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-one |
|---|
| InChI Identifier | InChI=1S/C5H8O5/c6-1-2-3(7)4(8)5(9)10-2/h2-4,6-8H,1H2/t2-,3-,4-/m1/s1 |
|---|
| InChI Key | CUOKHACJLGPRHD-BXXZVTAOSA-N |
|---|
| Isomeric SMILES | OC[C@H]1OC(=O)[C@H](O)[C@@H]1O |
|---|
| Average Molecular Weight | 148.114 |
|---|
| Monoisotopic Molecular Weight | 148.037173366 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as pentoses. These are monosaccharides in which the carbohydrate moiety contains five carbon atoms. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Pentoses |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pentose monosaccharide
- Gamma butyrolactone
- Tetrahydrofuran
- Carboxylic acid ester
- Lactone
- Secondary alcohol
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Oxacycle
- Organoheterocyclic compound
- Primary alcohol
- Hydrocarbon derivative
- Organic oxide
- Alcohol
- Carbonyl group
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 100 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | V6455 |
|---|
| Toronto Research Chemicals | R416000 |
|---|