| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:31:06 UTC |
|---|
| Update date | 2017-01-19 02:36:26 UTC |
|---|
| FoodComEx ID | PC000512 |
|---|
| FoodDB Record | FDB021867 |
|---|
| Chemical Information |
|---|
| Name | 2-Hydroxybutyric acid |
|---|
| Description | 2-Hydroxybutyric acid is an organic acid that is involved in propanoate metabolism. It is produced in mammalian tissues (principaly hepatic) that catabolize L-threonine or synthesize glutathione. Oxidative stress or detoxification demands can dramatically increase the rate of hepatic glutathione synthesis. Under such metabolic stress conditions, supplies of L-cysteine for glutathione synthesis become limiting, so homocysteine is diverted from the transmethylation pathway forming methionine into the transsulfuration pathway forming cystathionine. 2-Hydroxybutyrate is released as a by-product when cystathionine is cleaved to cysteine that is incorporated into glutathione. 2-Hydroxybutyric acid is often found in the urine of patients suffering from lactic acidosis and ketoacidosis. 2-Hydroxybutyric acid generally appears at high concentrations in situations related to deficient energy metabolism (e.g., birth asphyxia) and also in inherited metabolic diseases affecting the central nervous system during neonatal development, such as "cerebral" lactic acidosis, glutaric aciduria type II, dihydrolipoyl dehydrogenase (E3) deficiency, and propionic acidemia. More recently it has been noted that elevated levels of alpha-hydroxybutyrate in the plasma is a good marker for early stage type II diabetes (PMID: 19166731). It was concluded from studies done in the mid 1970's that an increased NADH2/NAD ratio was the most important factor for the production of 2-hydorxybutyric acid (PMID: 168632) [HMDB] |
|---|
| CAS Number | 600-15-7 |
|---|
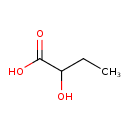
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (RS)-2-Hydroxybutyrate | hmdb | | (RS)-2-Hydroxybutyric acid | hmdb | | 2-hydroxy-Butanoate | hmdb | | 2-hydroxy-Butanoic acid | hmdb | | 2-hydroxy-DL-Butyrate | hmdb | | 2-hydroxy-DL-Butyric acid | hmdb | | 2-Hydroxy-n-butyrate | hmdb | | 2-Hydroxy-n-butyric acid | hmdb | | 2-Hydroxybutanoate | hmdb | | 2-Hydroxybutanoic acid | hmdb | | 2-Hydroxybutyrate | hmdb | | a-Hydroxy-n-butyrate | hmdb | | a-Hydroxy-n-butyric acid | hmdb | | a-Hydroxybutanoate | hmdb | | a-Hydroxybutanoic acid | hmdb | | a-Hydroxybutyrate | hmdb | | a-Hydroxybutyric acid | hmdb | | alpha-hydroxy-n-butyrate | hmdb | | alpha-hydroxy-n-butyric acid | hmdb | | alpha-Hydroxybutanoate | hmdb | | alpha-Hydroxybutanoic acid | hmdb | | alpha-Hydroxybutyrate | hmdb | | alpha-Hydroxybutyric acid | hmdb | | DL-2-Hydroxybutanoate | hmdb | | DL-2-Hydroxybutanoic acid | hmdb | | DL-a-Hydroxybutyrate | hmdb | | DL-a-Hydroxybutyric acid | hmdb | | DL-alpha-Hydroxybutyrate | hmdb | | DL-alpha-Hydroxybutyric acid | hmdb | | α-hydroxybutanoate | Generator | | α-hydroxybutanoic acid | Generator | | α-hydroxybutyrate | Generator | | α-hydroxybutyric acid | Generator |
|
|---|
| Chemical Formula | C4H8O3 |
|---|
| IUPAC name | 2-hydroxybutanoic acid |
|---|
| InChI Identifier | InChI=1S/C4H8O3/c1-2-3(5)4(6)7/h3,5H,2H2,1H3,(H,6,7) |
|---|
| InChI Key | AFENDNXGAFYKQO-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | CCC(O)C(O)=O |
|---|
| Average Molecular Weight | 104.1045 |
|---|
| Monoisotopic Molecular Weight | 104.047344122 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as alpha hydroxy acids and derivatives. These are organic compounds containing a carboxylic acid substituted with a hydroxyl group on the adjacent carbon. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Hydroxy acids and derivatives |
|---|
| Sub Class | Alpha hydroxy acids and derivatives |
|---|
| Direct Parent | Alpha hydroxy acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Fatty acid
- Alpha-hydroxy acid
- Secondary alcohol
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 10 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | Z2570 |
|---|
| MetaSci | HMDB0000008 |
|---|
| Sigma-Aldrich | HMDB0000008 |
|---|
| Toronto Research Chemicals | H831185 |
|---|