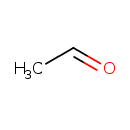
| Description | Acetaldehyde, also known as ethanal, belongs to the class of organic compounds known as short-chain aldehydes. These are aldehydes with a chain length containing between 2 and 5 carbon atoms. Acetaldehyde is an extremely weak basic (essentially neutral) compound (based on its pKa). Acetaldehyde exists in all living species, ranging from bacteria to humans. Within humans, acetaldehyde participates in a number of enzymatic reactions. In particular, acetaldehyde can be biosynthesized from ethanol; which is mediated by the enzyme alcohol dehydrogenase 1B. In addition, acetaldehyde can be converted into acetic acid; which is mediated by mitochondrial aldehyde dehydrogenases. Industrially, the main method of production of acetaldehyde is via the oxidation of ethylene via the Wacker process. In the 1970s, the world capacity of the Wacker-Hoechst direct oxidation process for producing acetaldehyde exceeded 2 million tonnes annually. Acetaldehyde is found in a number of different foods, such as sweet oranges, pineapples, and mandarin orange (clementine, tangerine), Acetaldehyde has also been detected, but not quantified in, several different foods, such as malabar plums, malus (crabapple), rose hips, natal plums, and medlars. This could make acetaldehyde a potential biomarker for the consumption of these foods. As a food constituent, acetaldehyde is an aldehydic, ethereal, and fruity tasting compound. Acetaldehyde is formally rated as a possible carcinogen (by IARC 2B) and is also a potentially toxic compound. Acetaldehyde has been found to be associated with several diseases such as alcoholism, ulcerative colitis, nonalcoholic fatty liver disease, and Crohn's disease. Acetaldehyde has also been linked to several inborn metabolic disorders including aldehyde dehydrogenase deficiency (III). The level at which an average consumer can detect acetaldehyde is still considerably lower than any toxicity. Pathways of exposure include air, water, land, or groundwater, as well as drink and smoke. Plastics Acetaldehyde is also created by thermal degradation or ultraviolet photo-degradation of some thermoplastic polymers during or after manufacture. Acetaldehyde is known to be the most abundant carcinogen in tobacco smoke; it is easily dissolved in saliva while smoking (PMID:17382522, PMID:21556207). |
|---|