| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:30:42 UTC |
|---|
| Update date | 2017-01-19 02:36:23 UTC |
|---|
| FoodComEx ID | PC000437 |
|---|
| FoodDB Record | FDB005416 |
|---|
| Chemical Information |
|---|
| Name | Guanidine |
|---|
| Description | Guanidine apparently acts by enhancing the release of acetylcholine following a nerve impulse. It also appears to slow the rates of depolarization and repolarization of muscle cell membranes.; Guanidine is a crystalline compound of strong alkalinity formed by the oxidation of guanine. It is used in the manufacture of plastics and explosives. It is found in urine as a normal product of protein metabolism. The molecule was first synthesized in 1861 by the oxidative degradation of an aromatic natural product, guanine, isolated from Peruvian guano. Despite the provocative simplicity of the molecule, the crystal structure was first described 148 years later.; Guanidine is a polyamine that can function as a strong organic base existing primarily as guanidium ions at physiological pH. With a pKa of 12.5, guanidine is protonated, with a charge of +1 in physiological conditions. It is found in the urine as a normal product of protein metabolism. It is also used in laboratory research as a protein denaturant. (From Martindale, the Extra Pharmacopoeia, 30th ed and Merck Index, 12th ed). Guanidine is a crystalline compound of strong alkalinity formed by the oxidation of guanine. It is used in the manufacture of plastics and explosives. -- Wikipedia; Guanidines are a group of organic compounds sharing a common functional group with the general structure (R1R2N)(R3R4N)C=N-R5. The central bond within this group is that of an imine; the other recognizable motif within this group is an aminal. Examples of guanidines are arginine, triazabicyclodecene and saxitoxin. other derivatives could include guanidine hydroxide, the active ingredient in some non-lye relaxers. Guanidinium salts are well known for their denaturing action on proteins. Guanidinium chloride is one of the most effective denaturants. In 6 M GndHCl all proteins with an ordered structure lose their structure, and most of them become randomly coiled, that is, they do not contain any residual structure. |
|---|
| CAS Number | 50-01-1 |
|---|
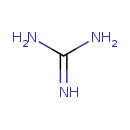
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (4-Aminobutyl) guanidine | HMDB | | (NH2)2C=NH | biospider | | 1184-68-5 (unspecified sulfate) | biospider | | 1763-07-1 (unspecified phosphate) | biospider | | 50-01-1 (mono-hydrochloride) | biospider | | Aminoformamidine | biospider | | Aminomethanamidine | biospider | | Bisguanidinium sulfate | biospider | | Carbamamidine | biospider | | Carbamidine | biospider | | GAI | biospider | | GU | biospider | | Guanidin | biospider | | Guanidine hydrochloride | biospider | | Guanidine, hydrochloride | biospider | | Guanidine, sulfate (2:1) | biospider | | Guanidinium sulfate | biospider | | H2N-C(=NH)-NH2 | biospider | | Imidourea | biospider | | Iminourea | biospider | | NSC7296 (SULFATE, 2:1) | biospider |
|
|---|
| Chemical Formula | CH5N3 |
|---|
| IUPAC name | guanidine |
|---|
| InChI Identifier | InChI=1S/CH5N3/c2-1(3)4/h(H5,2,3,4) |
|---|
| InChI Key | ZRALSGWEFCBTJO-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | NC(N)=N |
|---|
| Average Molecular Weight | 59.0705 |
|---|
| Monoisotopic Molecular Weight | 59.048347175 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as guanidines. Guanidines are compounds containing a guanidine moiety, with the general structure (R1R2N)(R3R4N)C=N-R5. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic nitrogen compounds |
|---|
| Class | Organonitrogen compounds |
|---|
| Sub Class | Guanidines |
|---|
| Direct Parent | Guanidines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Guanidine
- Carboximidamide
- Organopnictogen compound
- Hydrocarbon derivative
- Imine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | 182.3 oC | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 200 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| Not Available |