| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:30:28 UTC |
|---|
| Update date | 2017-01-19 02:36:22 UTC |
|---|
| FoodComEx ID | PC000405 |
|---|
| FoodDB Record | FDB022096 |
|---|
| Chemical Information |
|---|
| Name | 4,5-Dihydroorotic acid |
|---|
| Description | 4,5-Dihydroorotic acid is a substrate of the enzyme orotate reductase [EC 1.3.1.14], which is part of the pyrimidine metabolism pathway. (KEGG) Dihydroorotate is oxidized by Dihydroorotate dehydrogenases (DHODs) to orotate. These dehydrogenases use their FMN (flavin mononucleotide) prosthetic group to abstract a hydride equivalent from C6 to deprotonate C5 [HMDB] |
|---|
| CAS Number | 155-54-4 |
|---|
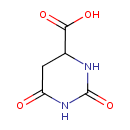
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (+)-2,6-dioxo-hexahydro-pyrimidine-4-carboxylate | hmdb | | (+)-2,6-dioxo-hexahydro-pyrimidine-4-carboxylic acid | hmdb | | (R)-2,6-dioxo-hexahydro-pyrimidine-4-carboxylate | hmdb | | (R)-2,6-dioxo-hexahydro-pyrimidine-4-carboxylic acid | hmdb | | (S)-2,6-dioxo-hexahydro-pyrimidine-4-carboxylate | hmdb | | (S)-2,6-dioxo-hexahydro-pyrimidine-4-carboxylic acid | hmdb | | 2,6-dioxo-hexahydro-pyrimidine-4-carboxylate | hmdb | | 2,6-dioxo-hexahydro-pyrimidine-4-carboxylic acid | hmdb | | 4,5-Dihydroorotate | hmdb | | 4,5-Dihydroorotic acid | hmdb | | 5,6-dihydro-Orotate | Generator | | 5,6-dihydro-Orotic acid | ChEBI | | 5,6-Dihydroorotate | hmdb | | DL-Dihydroortotate | Generator | | DL-Dihydroortotic acid | ChEBI | | Hydroorotate | Generator | | Hydroorotic acid | ChEBI | | L-hydroorotate | hmdb | | L-hydroorotic acid | hmdb | | R,S-Hydroorotate | hmdb |
|
|---|
| Chemical Formula | C5H6N2O4 |
|---|
| IUPAC name | 2,6-dioxo-1,3-diazinane-4-carboxylic acid |
|---|
| InChI Identifier | InChI=1S/C5H6N2O4/c8-3-1-2(4(9)10)6-5(11)7-3/h2H,1H2,(H,9,10)(H2,6,7,8,11) |
|---|
| InChI Key | UFIVEPVSAGBUSI-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | OC(=O)C1CC(=O)NC(=O)N1 |
|---|
| Average Molecular Weight | 158.1121 |
|---|
| Monoisotopic Molecular Weight | 158.03275669 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as alpha amino acids and derivatives. These are amino acids in which the amino group is attached to the carbon atom immediately adjacent to the carboxylate group (alpha carbon), or a derivative thereof. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Alpha amino acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-amino acid or derivatives
- N-acyl urea
- Pyrimidone
- Ureide
- 1,3-diazinane
- Pyrimidine
- Dicarboximide
- Urea
- Carbonic acid derivative
- Azacycle
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Organoheterocyclic compound
- Hydrocarbon derivative
- Organic oxide
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Organic oxygen compound
- Carbonyl group
- Organic nitrogen compound
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 5 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| Toronto Research Chemicals | D449970 |
|---|