| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:30:22 UTC |
|---|
| Update date | 2017-01-19 02:36:21 UTC |
|---|
| FoodComEx ID | PC000383 |
|---|
| FoodDB Record | FDB010505 |
|---|
| Chemical Information |
|---|
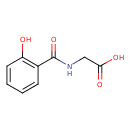
| Name | N-(2-Hydroxybenzoyl)glycine |
|---|
| Description | Constituent of milk
Salicyluric acid is an acyl glycine. Acyl glycines are normally minor metabolites of fatty acids. However, the excretion of certain acyl glycines is increased in several inborn errors of metabolism. In certain cases the measurement of these metabolites in body fluids can be used to diagnose disorders associated with mitochondrial fatty acid beta-oxidation. Acyl glycines are produced through the action of glycine N-acyltransferase (EC 2.3.1.13) which is an enzyme that catalyzes the chemical reaction:; acyl-CoA + glycine < -- > CoA + N-acylglycine; The induction of the salicyluric acid formation is one of the saturable pathways of salicylate elimination.; The formation of the methyl ester of salicyluric acid is observed during the quantitation of salicyluric acid and other salicylate metabolites in urine by high-pressure liquid chromatography. This methyl ester formation causes artificially low values for salicyluric acid and high values for salicylic acid.; (PMID: 6101164, 6857178). N-(2-Hydroxybenzoyl)glycine is found in milk and milk products. |
|---|
| CAS Number | 487-54-7 |
|---|
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| ((2-hydroxybenzoyl)amino)acetic acid | biospider | | (2-hydroxybenzamido)acetic acid | biospider | | (2-Hydroxybenzoyl)glycine | biospider | | [(2-Hydroxybenzoyl)amino]acetate | biospider | | [(2-Hydroxybenzoyl)amino]acetic acid | biospider | | 2-Hydroxybenzamidoacetic acid | db_source | | 2-hydroxybenzoylaminoacetate | biospider | | 2-hydroxybenzoylaminoacetic acid | biospider | | 2-Hydroxybenzoylglycine | biospider | | 2-Hydroxyhippurate | biospider | | 2-Hydroxyhippuric acid | biospider | | Glycine, N-(2-hydroxybenzoyl)- | biospider | | Glycine, N-(2-hydroxybenzoyl)- (9CI) | biospider | | Glycine, n-salicyloyl- | biospider | | Hippuric acid, o-hydroxy- | biospider | | N-(2-Hydroxybenzoyl)-glycine | biospider | | N-o-hydroxybenzoylglycine | biospider | | N-salicyloylglycine | biospider | | O-hydroxy-hippurate | biospider | | O-hydroxy-hippuric acid | biospider | | O-Hydroxyhippate | Generator | | O-Hydroxyhippic acid | Generator | | O-hydroxyhippurate | biospider | | O-hydroxyhippuric acid | biospider | | o-Hydroxyhippuric acid, 8CI | db_source | | Ortho-hydroxyhippurate | biospider | | Ortho-hydroxyhippuric acid | biospider | | Salicylglycine | biospider | | Salicyloylaminoacetic acid | db_source | | Salicyloylglycine | db_source | | Salicylurate | biospider | | Salicyluric acid | db_source |
|
|---|
| Chemical Formula | C9H9NO4 |
|---|
| IUPAC name | 2-[(2-hydroxyphenyl)formamido]acetic acid |
|---|
| InChI Identifier | InChI=1S/C9H9NO4/c11-7-4-2-1-3-6(7)9(14)10-5-8(12)13/h1-4,11H,5H2,(H,10,14)(H,12,13) |
|---|
| InChI Key | ONJSZLXSECQROL-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | OC(=O)CNC(=O)C1=CC=CC=C1O |
|---|
| Average Molecular Weight | 195.1721 |
|---|
| Monoisotopic Molecular Weight | 195.053157781 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as hippuric acids. Hippuric acids are compounds containing hippuric acid, which consists of a of a benzoyl group linked to the N-terminal of a glycine. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzoic acids and derivatives |
|---|
| Direct Parent | Hippuric acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hippuric acid
- N-acyl-alpha amino acid or derivatives
- N-acyl-alpha-amino acid
- Alpha-amino acid or derivatives
- Salicylic acid or derivatives
- Salicylamide
- Benzoyl
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Vinylogous acid
- Carboxamide group
- Secondary carboxylic acid amide
- Carboxylic acid derivative
- Carboxylic acid
- Monocarboxylic acid or derivatives
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Carbonyl group
- Organic oxygen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | 0.95 | HANSCH,C ET AL. (1995) |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Mp 170-172° | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 50 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| MetaSci | HMDB0000840 |
|---|
| Tokyo Chemical Industry | HMDB0000840 |
|---|
| Toronto Research Chemicals | H942885 |
|---|