| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:30:22 UTC |
|---|
| Update date | 2017-01-19 02:36:21 UTC |
|---|
| FoodComEx ID | PC000378 |
|---|
| FoodDB Record | FDB021920 |
|---|
| Chemical Information |
|---|
| Name | Pregnenolone |
|---|
| Description | Pregnenolone is a derivative of cholesterol, the product of Cytochrome P450 side-chain cleavage (EC 1.14.15.6, CYP11A1); this reaction consists of three consecutive monooxygenations; a 22-hydroxylation, 20-hydroxylation and the cleavage of the C20-C22 bond, yielding pregnenolone. Pregnenolone is the precursor to gonadal steroid hormones and the adrenal corticosteroids. This reaction occurs in steroid hormone-producing tissues such as the adrenal cortex, corpus luteum and placenta. The most notable difference between the placenta and other steroidogenic tissues is that electron supply to CYP11A1 limits the rate at which cholesterol is converted to pregnenolone in the placenta. The limiting component for electron delivery to CYP11A1 is the concentration of adrenodoxin reductase in the mitochondrial matrix which is insufficient to maintain the adrenodoxin pool in a fully reduced state. Pregnenolone is also a neurosteroid, and is produced in the spinal cord; CYP11A1 is the key enzyme catalyzing the conversion of cholesterol into pregnenolone, the rate-limiting step in the biosynthesis of all classes of steroids, and has been localized in sensory networks of the spinal cord dorsal horn. In the adrenal glomerulosa cell angiotensin II, one of the major physiological regulators of mineralocorticoid synthesis, appears to affect most of the cholesterol transfer to the mitochondrial outer membrane and transport to the inner membrane steps and thus to exerts a powerful control over the use of cholesterol for aldosterone production. (PMID: 17222962, 15823613, 16632873, 15134809) [HMDB]. Pregnenolone is found in many foods, some of which are common wheat, yellow bell pepper, oval-leaf huckleberry, and fenugreek. |
|---|
| CAS Number | 145-13-1 |
|---|
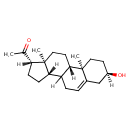
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (3b)-3-hydroxy-Pregn-5-en-20-one | hmdb | | 3-hydroxypregn-5-en-20-one | hmdb | | 3b-Hydroxypregn-5-en-20-one | hmdb | | 3beta-Hydroxypregn-5-en-20-one | hmdb | | 5-Pregnen-3b-ol-20-one | hmdb | | 5-Pregnen-3beta-ol-20-one | hmdb | | Natolone | hmdb | | Pregn-5-en-3b-ol-20-one | hmdb | | Pregn-5-ene-3b-ol-20-one | hmdb | | Pregnetan | hmdb | | Pregneton | hmdb | | Pregnolon | hmdb | | Prenolon | hmdb | | Regnosone | hmdb | | Skinostelon | hmdb |
|
|---|
| Chemical Formula | C21H32O2 |
|---|
| IUPAC name | 1-[(1S,2R,5R,11S,14S,15S)-5-hydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-7-en-14-yl]ethan-1-one |
|---|
| InChI Identifier | InChI=1S/C21H32O2/c1-13(22)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h4,15-19,23H,5-12H2,1-3H3/t15-,16?,17-,18+,19+,20+,21-/m1/s1 |
|---|
| InChI Key | ORNBQBCIOKFOEO-STZXPNGSSA-N |
|---|
| Isomeric SMILES | [H][C@@]12CC[C@H](C(C)=O)[C@@]1(C)CC[C@@]1([H])C2CC=C2C[C@H](O)CC[C@]12C |
|---|
| Average Molecular Weight | 316.4776 |
|---|
| Monoisotopic Molecular Weight | 316.240230268 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as gluco/mineralocorticoids, progestogins and derivatives. These are steroids with a structure based on a hydroxylated prostane moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Pregnane steroids |
|---|
| Direct Parent | Gluco/mineralocorticoids, progestogins and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Progestogin-skeleton
- 20-oxosteroid
- 3-hydroxy-delta-5-steroid
- 3-hydroxysteroid
- 3-alpha-hydroxysteroid
- Oxosteroid
- Hydroxysteroid
- Delta-5-steroid
- Cyclic alcohol
- Secondary alcohol
- Ketone
- Organic oxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Alcohol
- Organooxygen compound
- Organic oxide
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 100 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | F356 |
|---|
| AKSci | J10307 |
|---|
| AKSci | J90874 |
|---|
| Cayman Chemical | 19864 |
|---|
| MetaSci | HMDB0000253 |
|---|
| Sigma-Aldrich | HMDB0000253 |
|---|
| Toronto Research Chemicals | P712200 |
|---|