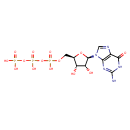| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:29:25 UTC |
|---|
| Update date | 2017-01-19 02:36:17 UTC |
|---|
| FoodComEx ID | PC000245 |
|---|
| FoodDB Record | FDB022527 |
|---|
| Chemical Information |
|---|
| Name | Guanosine triphosphate |
|---|
| Description | Guanosine triphosphate, also known as 5'-GTP or H4GTP, belongs to the class of organic compounds known as purine ribonucleoside triphosphates. These are purine ribobucleotides with a triphosphate group linked to the ribose moiety. Thus, a GTP-bound tubulin serves as a cap at the tip of microtubule to protect from depolymerization; and, once the GTP is hydrolyzed, the microtubule begins to depolymerize and shrink rapidly. Guanosine triphosphate is an extremely weak basic (essentially neutral) compound (based on its pKa). Guanosine triphosphate exists in all living species, ranging from bacteria to humans. In humans, guanosine triphosphate is involved in intracellular signalling through adenosine receptor A2B and adenosine. Guanosine-5'-triphosphate (GTP) is a purine nucleoside triphosphate. Outside of the human body, Guanosine triphosphate has been detected, but not quantified in, several different foods, such as mandarin orange (clementine, tangerine), coconuts, new zealand spinachs, sweet marjorams, and pepper (capsicum). This could make guanosine triphosphate a potential biomarker for the consumption of these foods. Cyclic guanosine triphosphate (cGTP) helps cyclic adenosine monophosphate (cAMP) activate cyclic nucleotide-gated ion channels in the olfactory system. It also has the role of a source of energy or an activator of substrates in metabolic reactions, like that of ATP, but more specific. It is used as a source of energy for protein synthesis and gluconeogenesis. For instance, a GTP molecule is generated by one of the enzymes in the citric acid cycle. GTP is also used as an energy source for the translocation of the ribosome towards the 3' end of the mRNA.During microtubule polymerization, each heterodimer formed by an alpha and a beta tubulin molecule carries two GTP molecules, and the GTP is hydrolyzed to GDP when the tubulin dimers are added to the plus end of the growing microtubule. The importing of these proteins plays an important role in several pathways regulated within the mitochondria organelle, such as converting oxaloacetate to phosphoenolpyruvate (PEP) in gluconeogenesis. GTP is involved in energy transfer within the cell. |
|---|
| CAS Number | 86-01-1 |
|---|
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| 5'-GTP | hmdb | | GTG | hmdb | | GTP | HMDB | | Guanosine 5'-(tetrahydrogen triate) | HMDB | | Guanosine 5'-(tetrahydrogen triphosphate) | hmdb | | Guanosine 5'-triate | ChEBI | | Guanosine 5'-triic acid | Generator | | Guanosine 5'-triorate | HMDB | | Guanosine 5'-trioric acid | ChEBI | | Guanosine 5'-triphosphate | hmdb | | Guanosine 5'-triphosphorate | hmdb | | Guanosine 5'-triphosphoric acid | hmdb | | Guanosine mono(tetrahydrogen triate) (ester) | HMDB | | Guanosine mono(tetrahydrogen triphosphate) (ester) | hmdb | | Guanosine triate | ChEBI | | Guanosine triic acid | Generator | | Guanosine Triphosphate | hmdb | | GUANOSINE-5'-triATE | ChEBI | | GUANOSINE-5'-triic acid | Generator | | H4gtp | hmdb |
|
|---|
| Chemical Formula | C10H16N5O14P3 |
|---|
| IUPAC name | ({[({[(2R,3S,4R,5R)-5-(2-amino-6-oxo-6,9-dihydro-1H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy}(hydroxy)phosphoryl)oxy](hydroxy)phosphoryl}oxy)phosphonic acid |
|---|
| InChI Identifier | InChI=1S/C10H16N5O14P3/c11-10-13-7-4(8(18)14-10)12-2-15(7)9-6(17)5(16)3(27-9)1-26-31(22,23)29-32(24,25)28-30(19,20)21/h2-3,5-6,9,16-17H,1H2,(H,22,23)(H,24,25)(H2,19,20,21)(H3,11,13,14,18)/t3-,5-,6-,9-/m1/s1 |
|---|
| InChI Key | XKMLYUALXHKNFT-UUOKFMHZSA-N |
|---|
| Isomeric SMILES | NC1=NC2=C(N=CN2[C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)C(=O)N1 |
|---|
| Average Molecular Weight | 523.1804 |
|---|
| Monoisotopic Molecular Weight | 522.990659781 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as purine ribonucleoside triphosphates. These are purine ribobucleotides with a triphosphate group linked to the ribose moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Nucleosides, nucleotides, and analogues |
|---|
| Class | Purine nucleotides |
|---|
| Sub Class | Purine ribonucleotides |
|---|
| Direct Parent | Purine ribonucleoside triphosphates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Purine ribonucleoside triphosphate
- Purine ribonucleoside monophosphate
- Pentose phosphate
- Pentose-5-phosphate
- Glycosyl compound
- N-glycosyl compound
- Monosaccharide phosphate
- Imidazopyrimidine
- Purine
- Monoalkyl phosphate
- Hydroxypyrimidine
- Alkyl phosphate
- Pyrimidine
- Monosaccharide
- Phosphoric acid ester
- N-substituted imidazole
- Organic phosphoric acid derivative
- Heteroaromatic compound
- Azole
- Imidazole
- Tetrahydrofuran
- Secondary alcohol
- 1,2-diol
- Oxacycle
- Organoheterocyclic compound
- Azacycle
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Alcohol
- Organic oxygen compound
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 10 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| Not Available |