| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:29:17 UTC |
|---|
| Update date | 2017-01-19 02:36:17 UTC |
|---|
| FoodComEx ID | PC000217 |
|---|
| FoodDB Record | FDB021808 |
|---|
| Chemical Information |
|---|
| Name | Dehydroepiandrosterone |
|---|
| Description | Dehydroepiandrosterone (DHEA) is a natural steroid hormone produced from cholesterol by the adrenal glands. DHEA is also produced in the gonads, adipose tissue and the brain. DHEA is structurally similar to, and is a precursor of, androstenedione, testosterone, estradiol, estrone and estrogen. It is the most abundant hormone in the human body. Most of DHEA is sulfated (dehydroepiandrosterone sulfate- DEHAS) before secretion. DHEAS is the sulfated version of DHEA; - this conversion is reversibly catalyzed by sulfotransferase (SULT2A1) primarily in the adrenals, the liver, and small intestines. In blood, most DHEA is found as DHEAS with levels that are about 300 times higher than free DHEA. Blood measurements of DHEAS/DHEA are useful to detect excess adrenal activity as seen in adrenal cancer or hyperplasia, including certain forms of congenital adrenal hyperplasia. Women with polycystic ovary syndrome tend to have normal or mildly elevated levels of DHEAS. [HMDB]. Dehydroepiandrosterone is found in many foods, some of which are summer grape, quinoa, calabash, and chinese chives. |
|---|
| CAS Number | 53-43-0 |
|---|
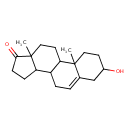
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (+)-Dehydroisoandrosterone | HMDB | | (3-beta)-3-Hydroxyandrost-5-en-17-one | HMDB | | (3beta,16alpha)-3,16-Dihydroxy-androst-5-en-17-one | HMDB | | (3beta)-3-Hydroxy-androst-5-en-17-one | HMDB | | 17-Chetovis | HMDB | | 17-Hormoforin | HMDB | | 3-beta-Hydroxy-5-androsten-17-one | HMDB | | 3-beta-Hydroxyandrost-5-en-17-one | HMDB | | 3b-Hydroxy-D5-androsten-17-one | HMDB | | 3b-Hydroxyandrost-5-en-17-one | HMDB | | 3beta-Hydroxy-5-androsten-17-one | HMDB | | 3beta-Hydroxy-androst-5-en-17-one | HMDB | | 3beta-Hydroxy-D5-androsten-17-one | HMDB | | 3beta-Hydroxyandrost-5-en-17-one | HMDB | | 5-Androsten-3-beta-ol-17-one | HMDB | | 5-Androsten-3b-ol-17-one | HMDB | | 5-Androsten-3beta-ol-17-one | HMDB | | 5-dehydro-Epiandrosterone | HMDB | | 5-Dehydroepiandrosterone | HMDB | | 5,6-Dehydroisoandrosterone | HMDB | | 5,6-Didehydroisoandrosterone | HMDB | | Andrestenol | HMDB | | Androst-5-ene-3b-ol-17-one | HMDB | | Androst-5-ene-3beta-ol-17-one | HMDB | | Androsten-3beta-ol-17-one | HMDB | | Androstenolone | HMDB | | Astenile | HMDB | | D5-Androsten-3b-ol-17-one | HMDB | | D5-Androsten-3beta-ol-17-one | HMDB | | Deandros | HMDB | | dehydro-Epi-androsterone | HMDB | | Dehydroisoandrosterone | HMDB | | Diandron | HMDB | | Diandrone | HMDB | | Hydroxyandrostenone | HMDB | | Prasterona | HMDB | | Prasterone | HMDB | | Prasteronum | HMDB | | Prestara | HMDB | | Psicosterone | HMDB | | trans-Dehydroandrosterone | HMDB |
|
|---|
| Chemical Formula | C19H28O2 |
|---|
| IUPAC name | 5-hydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-7-en-14-one |
|---|
| InChI Identifier | InChI=1S/C19H28O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h3,13-16,20H,4-11H2,1-2H3 |
|---|
| InChI Key | FMGSKLZLMKYGDP-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | CC12CCC3C(CC=C4CC(O)CCC34C)C1CCC2=O |
|---|
| Average Molecular Weight | 288.4244 |
|---|
| Monoisotopic Molecular Weight | 288.20893014 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as androgens and derivatives. These are 3-hydroxylated C19 steroid hormones. They are known to favor the development of masculine characteristics. They also show profound effects on scalp and body hair in humans. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Androstane steroids |
|---|
| Direct Parent | Androgens and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Androgen-skeleton
- 3-hydroxy-delta-5-steroid
- 3-hydroxysteroid
- Hydroxysteroid
- 17-oxosteroid
- Oxosteroid
- Delta-5-steroid
- Cyclic alcohol
- Ketone
- Secondary alcohol
- Organooxygen compound
- Alcohol
- Carbonyl group
- Organic oxygen compound
- Hydrocarbon derivative
- Organic oxide
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 3 g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | F793 |
|---|
| AKSci | J10065 |
|---|
| Cayman Chemical | 15728 |
|---|
| Toronto Research Chemicals | D229585 |
|---|
| Toronto Research Chemicals | KIT1095 |
|---|