| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:29:14 UTC |
|---|
| Update date | 2017-01-19 02:36:16 UTC |
|---|
| FoodComEx ID | PC000212 |
|---|
| FoodDB Record | FDB012163 |
|---|
| Chemical Information |
|---|
| Name | Dopamine |
|---|
| Description | Dopamine, also known as 3-hydroxytyramine or deoxyepinephrine, belongs to the class of organic compounds known as catecholamines and derivatives. Catecholamines and derivatives are compounds containing 4-(2-Aminoethyl)pyrocatechol [4-(2-aminoethyl)benzene-1,2-diol] or a derivative thereof formed by substitution. Dopamine is a drug which is used for the correction of hemodynamic imbalances present in the shock syndrome due to myocardial infarction, trauma, endotoxic septicemia, open-heart surgery, renal failure, and chronic cardiac decompensation as in congestive failure. Dopamine is a very strong basic compound (based on its pKa). Dopamine exists in all living organisms, ranging from bacteria to humans. Within humans, dopamine participates in a number of enzymatic reactions. In particular, dopamine can be biosynthesized from tyramine; which is catalyzed by the enzyme tyrosinase. In addition, dopamine and ascorbic acid can be converted into norepinephrine and dehydroascorbic acid; which is mediated by the enzyme dopamine beta-hydroxylase. Catechol in which the hydrogen at position 4 is substituted by a 2-aminoethyl group. In humans, dopamine is involved in disulfiram action pathway. Outside of the human body, Dopamine is found, on average, in the highest concentration within a few different foods, such as swiss chards, oats, and red beetroots and in a lower concentration in green zucchinis, broccoli, and yellow bell peppers. Dopamine has also been detected, but not quantified in, several different foods, such as potato, banana, avocado, eggplants, and custard apples. This could make dopamine a potential biomarker for the consumption of these foods. Dopamine is a potentially toxic compound. Dopamine, with regard to humans, has been found to be associated with several diseases such as bacterial meningitis, eosinophilic esophagitis, cerebral infarction, and schizophrenia; dopamine has also been linked to the inborn metabolic disorder aromatic l-amino acid decarboxylase deficiency. |
|---|
| CAS Number | 62-31-7 |
|---|
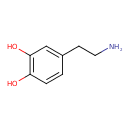
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (3H)-Dopamine | biospider | | 1,2-Benzenediol, 4-(2-aminoethyl)- (9CI) | biospider | | 2-(3,4-Dihydroxyphenyl)ethylamine | db_source | | 3-Hydroxtyramine | biospider | | 3-Hydroxytyramine | biospider | | 3,4-Dihydroxyphenethylamine | db_source | | 3,4-Dihydroxyphenylethylamine | biospider | | 4-(2-Aminoethyl)-1,2-benzenediol | ChEBI | | 4-(2-Aminoethyl)-1,2-benzenediol, 9CI | db_source | | 4-(2-Aminoethyl)-pyrocatechol | HMDB | | 4-(2-aminoethyl)benzene-1,2-diol | biospider | | 4-(2-Aminoethyl)catechol | biospider | | 4-(2-Aminoethyl)pyrocatechol | biospider | | a-(3,4-Dihydroxyphenyl)-b-aminoethane | biospider | | alpha-(3,4-Dihydroxyphenyl)-beta-aminoethane | biospider | | ASL 279 | db_source | | Cardiosteril | db_source | | Dopamin | biospider | | Dopamina | ChEBI | | Dopaminum | ChEBI | | Dopastat | db_source | | Dophamine | HMDB | | Dynatra | biospider | | Hydroxytyramin | biospider | | Hydroxytyramine | db_source | | Intropin | db_source | | NSC 169105 | db_source | | Oxytyramine | db_source | | Revivan | biospider |
|
|---|
| Chemical Formula | C8H11NO2 |
|---|
| IUPAC name | 4-(2-aminoethyl)benzene-1,2-diol |
|---|
| InChI Identifier | InChI=1S/C8H11NO2/c9-4-3-6-1-2-7(10)8(11)5-6/h1-2,5,10-11H,3-4,9H2 |
|---|
| InChI Key | VYFYYTLLBUKUHU-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | NCCC1=CC(O)=C(O)C=C1 |
|---|
| Average Molecular Weight | 153.1784 |
|---|
| Monoisotopic Molecular Weight | 153.078978601 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as catecholamines and derivatives. Catecholamines and derivatives are compounds containing 4-(2-Aminoethyl)pyrocatechol [4-(2-aminoethyl)benzene-1,2-diol] or a derivative thereof formed by substitution. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Phenols |
|---|
| Sub Class | Benzenediols |
|---|
| Direct Parent | Catecholamines and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Catecholamine
- Phenethylamine
- 2-arylethylamine
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Aralkylamine
- Monocyclic benzene moiety
- Amine
- Hydrocarbon derivative
- Primary amine
- Organopnictogen compound
- Organooxygen compound
- Organonitrogen compound
- Primary aliphatic amine
- Organic oxygen compound
- Organic nitrogen compound
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | -0.98 | HANSCH,C ET AL. (1995) |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | 128 oC | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 3 g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| Not Available |