| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:29:10 UTC |
|---|
| Update date | 2017-01-19 02:36:16 UTC |
|---|
| FoodComEx ID | PC000195 |
|---|
| FoodDB Record | FDB021798 |
|---|
| Chemical Information |
|---|
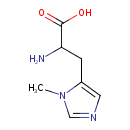
| Name | 3-Methyl-L-histidine |
|---|
| Description | 3-Methyl-L-histidine, also known as 3-methylhistidine, 3-MHis, 3MH, pi-methylhistidine or pros-methylhistidine, belongs to the class of organic compounds known as histidine and derivatives. 3MH is also classified as a methylamino acid. Methylamino acids are primarily proteogenic amino acids (found in proteins) which have been methylated (in situ) on their side chains by various methyltransferase enzymes. Histidine can be methylated at either the N1 or N3 position of its imidazole ring, yielding the isomers 1-methylhistidine (1MH; also referred to as tau-methylhistidine, according to IUPAC) or 3-methylhistidine (3MH; pi-methylhistidine, according to IUPAC), respectively. There is considerable confusion with regard to the nomenclature of the methylated nitrogen atoms on the imidazole ring of histidine in histidine-containing proteins (such as actin and myosin) as well as histidine-containing peptides (such as anserine and ophidine/balenine). In particular, older literature (mostly prior to the year 2000) as well as most biochemists and nutrition scientists incorrectly number the imidazole nitrogen atom most proximal to the side chain beta-carbon as 1 or N1, while organic chemists correctly designate it as 3 or N3. As a result, biochemists and nutrition scientists historically designated anserine (Npi methylated) as beta-alanyl-N1-methylhistidine (or beta-alanyl-1-methylhistidine), whereas according to standard IUPAC nomenclature, anserine is correctly named as beta-alanyl-N3-methylhistidine. As a result, for several decades, many papers incorrectly identified 1MH as a specific marker for dietary consumption or various pathophysiological effects when they really are referring to 3MH – and vice versa (PMID: 24137022 ). To help resolve this issue the IUPAC commission (PMID: 6743224 and IUPAC Compendium of Chemical Terminology, 2nd ed. (the "Gold Book"). Compiled by A. D. McNaught and A. Wilkinson. Blackwell Scientific Publications, Oxford (1997)) revised the nomenclature for histidine and introduced the terms pi (for prox or pros – near) and tau (for tele – far) to label the imidazole nitrogens in histidine. Therefore, the pi nitrogen is the nitrogen closest to the side chain beta carbon (atom #3 or N3) while the tau nitrogen is most distant from the side chain beta carbon (atom #1 or N1). IUPAC’s goal is for the global community to refer to the molecule depicted here is as “pi-methylhistidine” with the hope that the archaic term, 3-methylhistidine will eventually disappear. Unfortunately, this has not happened and confusion still persists. Older versions of the HMDB (prior to 2021) as well as current versions of PubChem, KEGG, UniProt and ChEBI indicate that an acceptable synonym for 3MH is tau-methylhistidine. This is incorrect and it continues to sow confusion. Indeed, a key paper that identified METTL18 as the enzyme responsible for tau-methylation of histidine incorrectly labelled the METTL18 product as 3MH (PMID: 33693809). Likewise, many members of the biochemical/nutrition community still incorrectly refer to 1MH as pi-methlyhistidine and 3MH as tau-methylhistidine. This has led to even more confusion. To maintain consistency for this compound description, all papers cited herein that incorrectly refer to 3MH as 1MH and vice versa, will have their conclusions re-stated and the citation will be marked with the phrase “3MH/1MH switch”. 3MH is a free amino acid arising from the proteolysis of 3MH-containing proteins and peptides. It is not synthesized on its own, nor can it be incorporated into proteins as an amino acid. However, it can be incorporated into certain dipeptides through the action of the enzyme known as Carnosine synthase I. 3MH can only be generated from histidine residues through the action of methyltransferases as a protein post-translational modification event. Histidine methylation on the 3- or pi site of histidine containing proteins is mediated by only one enzyme – METTL9. Recent discoveries have shown that 3MH is produced in essentially all vertebrates via the methyltransferase enzyme known as METTL9 (3MH/1MH switch - PMID: 33563959 ). METTL9 is a broad-specificity S-adenosylmethionine mediated methyltransferase that mediates the formation of the majority of 3MH present in mammalian and other vertebrate proteomes. METTL9-catalyzed methylation requires a His-x-His (HxH) motif, where "x" is a small amino acid consisting of A, N, G, S or T. This H[ANGST]H or HxH motif is found in a number of abundant mammalian proteins such as ARMC6, S100A9, and NDUFB3 (PMID: 33563959 ). In addition to these pi-His-methylated proteins, a specialized dipeptide called anserine (called beta-alanyl-3-methyl-L-histidine) that consists of beta-alanine and 3MH is also known and particularly well studied (PMID: 24137022). This methylated analog of carnosine, which is naturally produced in the liver via the enzyme Carnosine synthase I (PMID: 20097752), is especially abundant in the skeletal muscles and brains of mammals, birds and fish (PMID: 24137022 ). Anserine, like its homologs ophidine and carnosine, is believed to act as a pH buffer (for lactic acid generated by muscles), an antiglycating agent and an antioxidant. Neither ophidine nor anserine are produced in humans, with humans being the only vertebrate not producing methylated histidine versions of carnosine (PMID: 24137022 ). Because of its abundance in some muscle-related proteins but especially because of the high abundance of anserine found in poultry and fish, 3MH has been found to be a good biomarker for the consumption of meat (PMID: 21527577 ). Dietary studies have shown that general poultry consumption (p-trend = 0.0006) and especially chicken consumption (p-trend = 0.0003) are associated with increased levels of 3MH in human plasma (PMID: 30018457 ). The consumption of fish, especially salmon and cod, has also been shown to increase the levels of 3MH in serum and urine (3MH/1MH switch PMID: 31401679 ). As a general rule, urinary 3MH is associated with white meat intake (p< 0.001), whereas urinary 1MH is associated with red meat intake (p< 0.001) (3MH/1MH switch - PMID: 34091671). |
|---|
| CAS Number | 368-16-1 |
|---|
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (2S)-2-amino-3-(1-Methyl-1H-imidazol-5-yl)propanoate | Generator | | (2S)-2-amino-3-(1-Methyl-1H-imidazol-5-yl)propanoic acid | ChEBI | | 3-Methylhistidine | ChEBI | | 3-N-Methyl-L-histidine | HMDB | | L-3-Methylhistidine | HMDB | | N-pros-Methyl-L-histidine | ChEBI | | N(Pai)-methyl-L-histidine | ChEBI | | N(pros)-Methyl-L-histidine | HMDB | | N3-Methyl-L-histidine | HMDB | | Pi-methyl-L-histidine | HMDB | | Tau-methyl-L-histidine | HMDB | | Tau-methylhistidine | HMDB |
|
|---|
| Chemical Formula | C7H11N3O2 |
|---|
| IUPAC name | 2-amino-3-(1-methyl-1H-imidazol-5-yl)propanoic acid |
|---|
| InChI Identifier | InChI=1S/C7H11N3O2/c1-10-4-9-3-5(10)2-6(8)7(11)12/h3-4,6H,2,8H2,1H3,(H,11,12) |
|---|
| InChI Key | JDHILDINMRGULE-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | CN1C=NC=C1CC(N)C(O)=O |
|---|
| Average Molecular Weight | 169.1811 |
|---|
| Monoisotopic Molecular Weight | 169.085126611 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as histidine and derivatives. Histidine and derivatives are compounds containing cysteine or a derivative thereof resulting from reaction of cysteine at the amino group or the carboxy group, or from the replacement of any hydrogen of glycine by a heteroatom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Histidine and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Histidine or derivatives
- Alpha-amino acid
- Imidazolyl carboxylic acid derivative
- Aralkylamine
- N-substituted imidazole
- Azole
- Imidazole
- Heteroaromatic compound
- Amino acid
- Carboxylic acid
- Azacycle
- Organoheterocyclic compound
- Monocarboxylic acid or derivatives
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Primary amine
- Primary aliphatic amine
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Carbonyl group
- Organic oxygen compound
- Amine
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 80 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| MetaSci | HMDB0000479 |
|---|
| Sigma-Aldrich | HMDB0000479 |
|---|
| Toronto Research Chemicals | M312005 |
|---|