| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:29:07 UTC |
|---|
| Update date | 2017-01-19 02:36:15 UTC |
|---|
| FoodComEx ID | PC000185 |
|---|
| FoodDB Record | FDB022327 |
|---|
| Chemical Information |
|---|
| Name | S-(5'-Adenosyl)-L-homocysteine |
|---|
| Description | S-Adenosylhomocysteine, also known as adenosyl-homo-cys or adohcy, belongs to the class of organic compounds known as gamma butyrolactones. Gamma butyrolactones are compounds containing a gamma butyrolactone moiety, which consists of an aliphatic five-member ring with four carbon atoms, one oxygen atom, and bears a ketone group on the carbon adjacent to the oxygen atom. An organic sulfide that is the S-adenosyl derivative of L-homocysteine. S-Adenosylhomocysteine is a very strong basic compound (based on its pKa). S-Adenosylhomocysteine exists in all living species, ranging from bacteria to humans. Outside of the human body, S-Adenosylhomocysteine has been detected, but not quantified in, several different foods, such as arrowhead, guava, purslanes, kiwis, and gooseberries. This could make S-adenosylhomocysteine a potential biomarker for the consumption of these foods. S-Adenosylhomocysteine is a potentially toxic compound. |
|---|
| CAS Number | 979-92-0 |
|---|
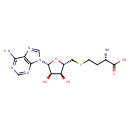
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (2S)-2-amino-4-({[(2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxytetrahydrofuran-2-yl]methyl}sulfanyl)butanoate | Generator | | (2S)-2-amino-4-({[(2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxytetrahydrofuran-2-yl]methyl}sulfanyl)butanoic acid | ChEBI | | (2S)-2-amino-4-({[(2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxytetrahydrofuran-2-yl]methyl}sulphanyl)butanoate | Generator | | (2S)-2-amino-4-({[(2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxytetrahydrofuran-2-yl]methyl}sulphanyl)butanoic acid | Generator | | (S)-5'-(S)-(3-Amino-3-carboxypropyl)-5'-thioadenosine | hmdb | | 2-S-adenosyl-L-homocysteine | hmdb | | 5'-Deoxy-S-adenosyl-L-homocysteine | hmdb | | 5'-S-(3-amino-3-carboxypropyl)-5'-thio-L-Adenosine | hmdb | | Adenosyl-homo-CYS | hmdb | | Adenosyl-L-homocysteine | hmdb | | Adenosylhomo-CYS | hmdb | | Adenosylhomocysteine | hmdb | | AdoHcy | ChEBI | | Formycinylhomocysteine | hmdb | | L-5'-S-(3-amino-3-carboxypropyl)-5'-thior-Adenosine | hmdb | | L-S-adenosyl-Homocysteine | hmdb | | L-S-Adenosylhomocysteine | hmdb | | S-(5'-Adenosyl)-L-homocysteine | hmdb | | S-(5'-Deoxyadenosin-5'-yl)-L-homocysteine | hmdb | | S-(5'-Deoxyadenosine-5')-L-homocysteine | HMDB | | S-[1-(Adenin-9-yl)-1,5-dideoxy-b-D-ribofuranos-5-yl]-L-homocysteine | Generator | | S-[1-(Adenin-9-yl)-1,5-dideoxy-beta-D-ribofuranos-5-yl]-L-homocysteine | ChEBI | | S-[1-(Adenin-9-yl)-1,5-dideoxy-β-D-ribofuranos-5-yl]-L-homocysteine | Generator | | S-Adenosyl-homocysteine | hmdb | | S-Adenosyl-L-homocysteine | hmdb | | S-Adenosylhomocysteine | ChEBI | | SAH | ChEBI |
|
|---|
| Chemical Formula | C14H20N6O5S |
|---|
| IUPAC name | (2S)-2-amino-4-({[(2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methyl}sulfanyl)butanoic acid |
|---|
| InChI Identifier | InChI=1S/C14H20N6O5S/c15-6(14(23)24)1-2-26-3-7-9(21)10(22)13(25-7)20-5-19-8-11(16)17-4-18-12(8)20/h4-7,9-10,13,21-22H,1-3,15H2,(H,23,24)(H2,16,17,18)/t6-,7+,9+,10+,13+/m0/s1 |
|---|
| InChI Key | ZJUKTBDSGOFHSH-WFMPWKQPSA-N |
|---|
| Isomeric SMILES | N[C@@H](CCSC[C@H]1O[C@H]([C@H](O)[C@@H]1O)N1C=NC2=C1N=CN=C2N)C(O)=O |
|---|
| Average Molecular Weight | 384.411 |
|---|
| Monoisotopic Molecular Weight | 384.12158847 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as gamma butyrolactones. Gamma butyrolactones are compounds containing a gamma butyrolactone moiety, which consists of an aliphatic five-member ring with four carbon atoms, one oxygen atom, and bears a ketone group on the carbon adjacent to the oxygen atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Lactones |
|---|
| Sub Class | Gamma butyrolactones |
|---|
| Direct Parent | Gamma butyrolactones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Gamma butyrolactone
- Tetrahydrofuran
- Secondary alcohol
- Carboxylic acid ester
- 1,2-diol
- Oxacycle
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 10 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | X6831 |
|---|
| Cayman Chemical | 13603 |
|---|
| Toronto Research Chemicals | A291500 |
|---|