| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:28:07 UTC |
|---|
| Update date | 2017-01-19 02:36:12 UTC |
|---|
| FoodComEx ID | PC000085 |
|---|
| FoodDB Record | FDB001498 |
|---|
| Chemical Information |
|---|
| Name | Glutathione |
|---|
| Description | Glutathione, also known as GSH or agifutol S, belongs to the class of organic compounds known as peptides. Peptides are compounds containing an amide derived from two or more amino carboxylic acid molecules (the same or different) by formation of a covalent bond from the carbonyl carbon of one to the nitrogen atom of another. Glutathione is a drug which is used for nutritional supplementation, also for treating dietary shortage or imbalance. Glutathione is a very strong basic compound (based on its pKa). A tripeptide compound consisting of glutamic acid attached via its side chain to the N-terminus of cysteinylglycine. Glutathione exists in all living species, ranging from bacteria to humans. Within humans, glutathione participates in a number of enzymatic reactions. In particular, leukotriene A4 and glutathione can be converted into leukotriene C4 through its interaction with the enzyme leukotriene C4 synthase. In addition, 15(S)-hpete and glutathione can be converted into 15(S)-hete and oxidized glutathione through its interaction with the enzyme glutathione peroxidase 1. In humans, glutathione is involved in mefenamic acid action pathway. Outside of the human body, Glutathione is found, on average, in the highest concentration within a few different foods, such as purslanes, spinachs, and corns. Glutathione has also been detected, but not quantified in, several different foods, such as yautia, coconuts, common salsifies, pigeon pea, and ohelo berries. This could make glutathione a potential biomarker for the consumption of these foods. Glutathione is a potentially toxic compound. |
|---|
| CAS Number | 70-18-8 |
|---|
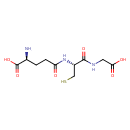
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| 5-L-Glutamyl-L-cysteinylglycine | biospider | | Agifutol S | HMDB | | Bakezyme rx | HMDB | | Copren | HMDB | | Deltathione | HMDB | | g-L-Glutamyl-L-cysteinyl-glycine | Generator | | gamma-Glutamylcysteinylglycine | biospider | | gamma-L-Glutamyl-L-cysteinyl-glycine | biospider | | gamma-L-Glutamyl-L-cysteinylglycine | HMDB | | Glutathion | HMDB | | Glutathione | biospider | | Glutathione (reduced) | biospider | | Glutathione red | HMDB | | Glutathione reduced | HMDB | | Glutathione-SH | biospider | | Glutathione, reduced form | biospider | | Glutatiol | HMDB | | Glutatione | HMDB | | Glutide | HMDB | | Glutinal | HMDB | | GSH | manual | | Isethion | HMDB | | L-g-Glutamyl-L-cysteinyl-glycine | biospider | | L-gamma-Glutamyl-L-cysteinyl-glycine | biospider | | L-gamma-Glutamyl-L-cysteinylglycine | HMDB | | L-Glutamyl-L-cysteinylglycine | HMDB | | L-Glutathione | db_source | | L-Glutathione reduce | HMDB | | L-Glutathione reduced | biospider | | Ledac | HMDB | | N-(N-g-L-Glutamyl-L-cysteinyl)glycine | Generator | | N-(N-gamma-L-Glutamyl-L-cysteinyl)glycine | biospider | | N-(N-L-gamma-Glutamyl-L-cysteinyl)glycine | biospider | | N-(N-γ-L-glutamyl-L-cysteinyl)glycine | Generator | | Neuthion | HMDB | | Red. glutathione | HMDB | | Reduced glutathione | biospider | | Tathion | HMDB | | Tathione | HMDB | | Triptide | HMDB | | γ-L-glutamyl-L-cysteinyl-glycine | Generator |
|
|---|
| Chemical Formula | C10H17N3O6S |
|---|
| IUPAC name | (2S)-2-amino-4-{[(1R)-1-[(carboxymethyl)carbamoyl]-2-sulfanylethyl]carbamoyl}butanoic acid |
|---|
| InChI Identifier | InChI=1S/C10H17N3O6S/c11-5(10(18)19)1-2-7(14)13-6(4-20)9(17)12-3-8(15)16/h5-6,20H,1-4,11H2,(H,12,17)(H,13,14)(H,15,16)(H,18,19)/t5-,6-/m0/s1 |
|---|
| InChI Key | RWSXRVCMGQZWBV-WDSKDSINSA-N |
|---|
| Isomeric SMILES | N[C@@H](CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O)C(O)=O |
|---|
| Average Molecular Weight | 307.323 |
|---|
| Monoisotopic Molecular Weight | 307.083805981 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as peptides. Peptides are compounds containing an amide derived from two or more amino carboxylic acid molecules (the same or different) by formation of a covalent bond from the carbonyl carbon of one to the nitrogen atom of another. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Peptides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha peptide
- N-acyl-alpha-amino acid
- N-acyl-alpha amino acid or derivatives
- Alpha-amino acid
- Alpha-amino acid or derivatives
- L-alpha-amino acid
- Dicarboxylic acid or derivatives
- Fatty acid
- Amino acid or derivatives
- Amino acid
- Alkylthiol
- Carboximidic acid
- Carboximidic acid derivative
- Carboxylic acid
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Hydrocarbon derivative
- Primary aliphatic amine
- Organic oxide
- Organic oxygen compound
- Organic nitrogen compound
- Carbonyl group
- Amine
- Organonitrogen compound
- Organooxygen compound
- Organosulfur compound
- Organopnictogen compound
- Primary amine
- Aliphatic acyclic compound
|
|---|
| Molecular Framework | Aliphatic acyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | 195 oC | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 4 g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | J10806 |
|---|
| AKSci | J91462 |
|---|
| AKSci | X7257 |
|---|
| AKSci | HMDB0000125 |
|---|
| Cayman Chemical | 10007461 |
|---|
| Glentham | GE1520 |
|---|
| MetaSci | HMDB0000125 |
|---|
| Toronto Research Chemicals | G597951 |
|---|