| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:28:02 UTC |
|---|
| Update date | 2017-01-19 02:36:12 UTC |
|---|
| FoodComEx ID | PC000078 |
|---|
| FoodDB Record | FDB021889 |
|---|
| Chemical Information |
|---|
| Name | Epinephrine |
|---|
| Description | Epinephrine is a catecholamine, a sympathomimetic monoamine derived from the amino acids phenylalanine and tyrosine. It is the active sympathomimetic hormone secreted from the adrenal medulla in most species. It stimulates both the alpha- and beta- adrenergic systems, causes systemic vasoconstriction and gastrointestinal relaxation, stimulates the heart, and dilates bronchi and cerebral vessels. It is used in asthma and cardiac failure and to delay absorption of local anesthetics. Epinephrine also constricts arterioles in the skin and gut while dilating arterioles in leg muscles. It elevates the blood sugar level by increasing hydrolysis of glycogen to glucose in the liver, and at the same time begins the breakdown of lipids in adipocytes. Epinephrine has a suppressive effect on the immune system. [HMDB] |
|---|
| CAS Number | 51-43-4 |
|---|
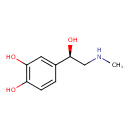
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (-)-(R)-Epinephrine | hmdb | | (-)-3,4-Dihydroxy-a-((methylamino)methyl)benzyl alcohol | Generator | | (-)-3,4-dihydroxy-a-[(methylamino)methyl]-Benzyl alcohol | hmdb | | (-)-3,4-Dihydroxy-a-[2-(methylamino)ethyl]benzyl alcohol | hmdb | | (-)-3,4-Dihydroxy-alpha-((methylamino)methyl)benzyl alcohol | ChEBI | | (-)-3,4-dihydroxy-alpha-[(methylamino)methyl]-Benzyl alcohol | hmdb | | (-)-3,4-Dihydroxy-alpha-[2-(methylamino)ethyl]benzyl alcohol | hmdb | | (-)-3,4-Dihydroxy-α-((methylamino)methyl)benzyl alcohol | Generator | | (-)-Adrenaline | hmdb | | (-)-Epinephrine | hmdb | | (R)-(-)-Adnephrine | ChEBI | | (R)-(-)-Adrenaline | ChEBI | | (R)-(-)-Epinephrine | ChEBI | | (R)-(-)-Epirenamine | ChEBI | | (R)-4-[1-hydroxy-2-(methylamino)ethyl]-1,2-Benzenediol | hmdb | | (R)-Adrenaline | hmdb | | (R)-Epinephrine | hmdb | | 4-[(1R)-1-hydroxy-2-(methylamino)ethyl]-1,2-Benzenediol | hmdb | | Adnephrine | hmdb | | Adrenal | hmdb | | Adrenalin | hmdb | | Adrenaline | hmdb | | Adrenine | hmdb | | Adrin | hmdb | | Ana-Kit | hmdb | | Bosmin | hmdb | | Bronkaid Mist | hmdb | | Chelafrin | hmdb | | Epifrin | hmdb | | Epiglaufrin | hmdb | | Epinefrina | hmdb | | Epinephran | hmdb | | Epinephrin | ChEBI | | Epinephrine | hmdb | | Epinephrinum | ChEBI | | Epipen | hmdb | | Epipen jr | ChEBI | | Epirenan | hmdb | | Eppy | hmdb | | Exadrin | hmdb | | Glauposine | hmdb | | Hemisine | hmdb | | Hemostasin | hmdb | | Hemostatin | hmdb | | Hypernephrin | hmdb | | Isoptoepinal | hmdb | | l-1-(3,4-Dihydroxyphenyl)-2-methylaminoethanol | hmdb | | L-Adrenaline | hmdb | | l-Epinephrine | hmdb | | l-Epirenamine | hmdb | | L-Methylaminoethanolcatechol | hmdb | | Levoepinephrine | hmdb | | Levorenen | hmdb | | Levorenin | hmdb | | Levorenine | hmdb | | Levoreninum | hmdb | | Lyodrin | hmdb | | Methylarterenol | hmdb | | Mucidrina | hmdb | | Nephridine | hmdb | | Nieraline | hmdb | | Paranephrin | hmdb | | Primatene | ChEBI | | Primatene Mist | hmdb | | R-(-)-Epinephrine | hmdb | | Renaglandin | hmdb | | Renaleptine | hmdb | | Renalina | hmdb | | Renoform | hmdb | | Renostypticin | hmdb | | Renostyptin | hmdb | | Scurenaline | hmdb | | Simplene | hmdb | | Styptirenal | hmdb | | Supracapsulin | hmdb | | Supranephrane | hmdb | | Suprarenaline | hmdb | | Suprarenin | hmdb | | Surrenine | hmdb | | Sus-phrine | hmdb | | Takamina | hmdb | | Vasoconstrictine | hmdb | | Vasotonin | hmdb |
|
|---|
| Chemical Formula | C9H13NO3 |
|---|
| IUPAC name | 4-[(1R)-1-hydroxy-2-(methylamino)ethyl]benzene-1,2-diol |
|---|
| InChI Identifier | InChI=1S/C9H13NO3/c1-10-5-9(13)6-2-3-7(11)8(12)4-6/h2-4,9-13H,5H2,1H3/t9-/m0/s1 |
|---|
| InChI Key | UCTWMZQNUQWSLP-VIFPVBQESA-N |
|---|
| Isomeric SMILES | CNC[C@H](O)C1=CC(O)=C(O)C=C1 |
|---|
| Average Molecular Weight | 183.2044 |
|---|
| Monoisotopic Molecular Weight | 183.089543287 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as catechols. Catechols are compounds containing a 1,2-benzenediol moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Phenols |
|---|
| Sub Class | Benzenediols |
|---|
| Direct Parent | Catechols |
|---|
| Alternative Parents | |
|---|
| Substituents | - Catechol
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Aralkylamine
- Monocyclic benzene moiety
- 1,2-aminoalcohol
- Secondary alcohol
- Secondary amine
- Secondary aliphatic amine
- Aromatic alcohol
- Alcohol
- Organooxygen compound
- Organonitrogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Amine
- Organopnictogen compound
- Hydrocarbon derivative
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 1 g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | J10365 |
|---|
| AKSci | L926 |
|---|
| Cayman Chemical | 18626 |
|---|
| Glentham | GP2165 |
|---|
| MetaSci | HMDB0000068 |
|---|
| Sigma-Aldrich | HMDB0000068 |
|---|
| Toronto Research Chemicals | E588580 |
|---|