| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:28:01 UTC |
|---|
| Update date | 2017-01-19 02:36:12 UTC |
|---|
| FoodComEx ID | PC000077 |
|---|
| FoodDB Record | FDB001936 |
|---|
| Chemical Information |
|---|
| Name | Stigmasterol |
|---|
| Description | Stigmasterol, also known as poriferasterol or phytosterol, belongs to the class of organic compounds known as stigmastanes and derivatives. These are sterol lipids with a structure based on the stigmastane skeleton, which consists of a cholestane moiety bearing an ethyl group at the carbon atom C24. Stigmasterol is a phytosterol, meaning it is steroid derived from plants. ,It is very hydrophobic, practically insoluble in water, and relatively neutral. Stigmasterol is among the most abundant of plant sterols with its major function being to maintain the structure and physiology of cell membranes. It is found in plant fats or oils associated with various vegetables, legumes, nuts, and seeds such as soybean, calabar bean, rape seed and various herbs. Stigmasterol is also a constituent of unpasteurized milk. Pasteurization is known to inactivate stigmasterol. Stigmasterol is a food additive in manufactured food products in the United Kingdom and European Union. One such phytosterol complex, isolated from vegetable oil, is cholestatin, composed of campesterol, stigmasterol, and brassicasterol, and is marketed as a dietary supplement. Consumption of 2 grams per day of plant sterols is associated with a reduction in blood LDL cholesterol of 8-10% (PMID: 29267628). As the typical western diet contains only about 300 mg/day of phytosterols, foods enriched with phytosterols are usually used to achieve the recommended intake (PMID: 29267628). The mechanism behind phytosterols lowering cholesterol involves the inhibition of the incorporation of cholesterol into micelles in the gastrointestinal tract, which decreases the overall amount of cholesterol absorbed. This may in turn help to control body total plasma cholesterol levels, as well as modify HDL, LDL and TAG levels in blood. Many margarines, butters, breakfast cereals and spreads are now enriched with phytosterols and marketed towards people wishing to lower their cholesterol levels. More recently, stigmasterol has been identified as a potent and independent risk factor for cardiovascular disease. Accumulation of stigmasterol leads to left ventricle dysfunction, cardiac interstitial fibrosis and macrophage infiltration without atherosclerosis (PMID: 30675518). Stigmasterol has been found to be associated with phytosterolemia, a disease characterized by elevated levels of dietary plant sterols in the blood. (PMID: 30675518). |
|---|
| CAS Number | 83-48-7 |
|---|
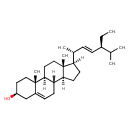
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (22E)-Stigmasta-5,22-dien-3-ol | biospider | | (22E)-Stigmasta-5,22-dien-3beta-ol | biospider | | (24S)-24-Ethylcholesta-5,22-dien-3beta-ol | biospider | | (24S)-5,22-Stigmastadien-3β-ol | biospider | | (24S)-5,22-stigmastadien-3b-ol | biospider | | (24S)-5,22-Stigmastadien-3beta-ol | biospider | | (24S)-Stigmast-5,22-dien-3beta-ol | biospider | | (3b,22E)-Stigmasta-5,22-dien-3-ol | biospider | | (3beta,22E)-Stigmasta-5,22-dien-3-ol | biospider | | β-Stigmasterol | biospider | | b-Stigmasterol | biospider | | beta-Stigmasterol | biospider | | D5-Stigmasterol | biospider | | Delta5-Stigmasterol | biospider | | Delta5,22-Stigmastadien-3beta-ol | biospider | | Serposterol | db_source | | Stigmasta-5,22-dien-3-b-ol | biospider | | Stigmasta-5,22-dien-3-beta-ol | biospider | | Stigmasta-5,22-dien-3-ol, (3β,22E)- | biospider | | Stigmasta-5,22-dien-3-ol, (3β)- | biospider | | Stigmasta-5,22-dien-3-ol, (3beta,22E)- (9CI) | biospider | | Stigmasta-5,22-dien-3-ol, (3beta)- | biospider | | Stigmasta-5,22-dien-3β-ol | biospider | | Stigmasta-5,22-dien-3beta-ol (8CI) | biospider | | Stigmasterin | biospider | | Stigmasterol | db_source |
|
|---|
| Chemical Formula | C29H48O |
|---|
| IUPAC name | (1S,2R,5S,10S,11S,14R,15R)-14-[(2R,3E,5S)-5-ethyl-6-methylhept-3-en-2-yl]-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-7-en-5-ol |
|---|
| InChI Identifier | InChI=1S/C29H48O/c1-7-21(19(2)3)9-8-20(4)25-12-13-26-24-11-10-22-18-23(30)14-16-28(22,5)27(24)15-17-29(25,26)6/h8-10,19-21,23-27,30H,7,11-18H2,1-6H3/b9-8+/t20-,21-,23+,24+,25-,26+,27+,28+,29-/m1/s1 |
|---|
| InChI Key | HCXVJBMSMIARIN-PHZDYDNGSA-N |
|---|
| Isomeric SMILES | [H][C@@]1(CC[C@@]2([H])[C@]3([H])CC=C4C[C@@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)\C=C\[C@@H](CC)C(C)C |
|---|
| Average Molecular Weight | 412.702 |
|---|
| Monoisotopic Molecular Weight | 412.370516166 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as stigmastanes and derivatives. These are sterol lipids with a structure based on the stigmastane skeleton, which consists of a cholestane moiety bearing an ethyl group at the carbon atom C24. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Stigmastanes and derivatives |
|---|
| Direct Parent | Stigmastanes and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - C24-propyl-sterol-skeleton
- Stigmastane-skeleton
- Triterpenoid
- 3-hydroxy-delta-5-steroid
- 3-hydroxysteroid
- Hydroxysteroid
- 3-beta-hydroxysteroid
- 3-beta-hydroxy-delta-5-steroid
- Delta-5-steroid
- Cyclic alcohol
- Secondary alcohol
- Alcohol
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxygen compound
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Mp 170° | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 2 g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | P501 |
|---|
| AKSci | HMDB0000937 |
|---|
| Cayman Chemical | 18079 |
|---|
| Glentham | GP9119 |
|---|
| Glentham | GP5909 |
|---|
| MetaSci | HMDB0000937 |
|---|
| Toronto Research Chemicals | S686750 |
|---|