| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:28:01 UTC |
|---|
| Update date | 2017-01-19 02:36:12 UTC |
|---|
| FoodComEx ID | PC000076 |
|---|
| FoodDB Record | FDB022200 |
|---|
| Chemical Information |
|---|
| Name | Kynurenic acid |
|---|
| Description | Kynurenic acid (KYNA) is a well-known endogenous antagonist of the glutamate ionotropic excitatory amino acid receptors N-methyl-D-aspartate (NMDA), alphaamino-3-hydroxy-5-methylisoxazole-4-propionic acid and kainate receptors and of the nicotine cholinergic subtype alpha 7 receptors. KYNA neuroprotective and anticonvulsive activities have been demonstrated in animal models of neurodegenerative diseases. Because of KYNA's neuromodulatory character, its involvement has been speculatively linked to the pathogenesis of a number of neurological conditions including those in the ageing process. Different patterns of abnormalities in various stages of KYNA metabolism in the CNS have been reported in Alzheimer's disease, Parkinson's disease and Huntington's disease. In HIV-1-infected patients and in patients with Lyme neuroborreliosis a marked rise of KYNA metabolism was seen. In the ageing process KYNA metabolism in the CNS of rats shows a characteristic pattern of changes throughout the life span. A marked increase of the KYNA content in the CNS occurs before the birth, followed by a dramatic decline on the day of birth. A low activity was seen during ontogenesis, and a slow and progressive enhancement occurs during maturation and ageing. This remarkable profile of KYNA metabolism alterations in the mammalian brain has been suggested to result from the development of the organisation of neuronal connections and synaptic plasticity, development of receptor recognition sites, maturation and ageing. There is significant evidence that KYNA can improve cognition and memory, but it has also been demonstrated that it interferes with working memory. Impairment of cognitive function in various neurodegenerative disorders is accompanied by profound reduction and/or elevation of KYNA metabolism. The view that enhancement of CNS KYNA levels could underlie cognitive decline is supported by the increased KYNA metabolism in Alzheimer's disease, by the increased KYNA metabolism in down's syndrome and the enhancement of KYNA function during the early stage of Huntington's disease. Kynurenic acid is the only endogenous N-methyl-D-aspartate (NMDA) receptor antagonist identified up to now, that mediates glutamatergic hypofunction. Schizophrenia is a disorder of dopaminergic neurotransmission, but modulation of the dopaminergic system by glutamatergic neurotransmission seems to play a key role. Despite the NMDA receptor antagonism, kynurenic acid also blocks, in lower doses, the nicotinergic acetycholine receptor, i.e., increased kynurenic acid levels can explain psychotic symptoms and cognitive deterioration. Kynurenic acid levels are described to be higher in the cerebrospinal fluid (CSF) and in critical central nervous system (CNS) regions of schizophrenics as compared to controls. (PMID: 17062375, 16088227) [HMDB] |
|---|
| CAS Number | 492-27-3 |
|---|
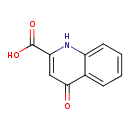
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| 2-Carboxy-4-hydroxyquinoline | hmdb | | 4-Hydroxy-2-chinolincarbonsaeure | ChEBI | | 4-Hydroxy-2-quinolinecarboxylate | Generator | | 4-hydroxy-2-Quinolinecarboxylic acid | hmdb | | 4-hydroxy-Quinaldate | hmdb | | 4-hydroxy-Quinaldic acid | hmdb | | 4-Hydroxyquinaldate | hmdb | | 4-Hydroxyquinaldic acid | hmdb | | 4-Hydroxyquinaldinate | hmdb | | 4-Hydroxyquinaldinic acid | hmdb | | 4-Hydroxyquinoline-2-carboxylate | hmdb | | 4-Hydroxyquinoline-2-carboxylic acid | hmdb | | Kynurenate | hmdb | | Kynurenic acid | hmdb | | Kynurensaeure | ChEBI | | Quinurenic acid | hmdb |
|
|---|
| Chemical Formula | C10H7NO3 |
|---|
| IUPAC name | 4-oxo-1,4-dihydroquinoline-2-carboxylic acid |
|---|
| InChI Identifier | InChI=1S/C10H7NO3/c12-9-5-8(10(13)14)11-7-4-2-1-3-6(7)9/h1-5H,(H,11,12)(H,13,14) |
|---|
| InChI Key | HCZHHEIFKROPDY-UHFFFAOYSA-N |
|---|
| Isomeric SMILES | OC(=O)C1=CC(=O)C2=CC=CC=C2N1 |
|---|
| Average Molecular Weight | 189.1675 |
|---|
| Monoisotopic Molecular Weight | 189.042593095 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as quinoline carboxylic acids. These are quinolines in which the quinoline ring system is substituted by a carboxyl group at one or more positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Quinolines and derivatives |
|---|
| Sub Class | Quinoline carboxylic acids |
|---|
| Direct Parent | Quinoline carboxylic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Quinoline-2-carboxylic acid
- Dihydroquinolone
- Dihydroquinoline
- Pyridine carboxylic acid
- Pyridine carboxylic acid or derivatives
- Pyridine
- Benzenoid
- Heteroaromatic compound
- Vinylogous amide
- Monocarboxylic acid or derivatives
- Carboxylic acid
- Azacycle
- Carboxylic acid derivative
- Hydrocarbon derivative
- Organic oxide
- Organopnictogen compound
- Organooxygen compound
- Organonitrogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, up to 1 g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | W6496 |
|---|
| MetaSci | HMDB0000715 |
|---|
| Sigma-Aldrich | HMDB0000715 |
|---|
| Toronto Research Chemicals | K660500 |
|---|