| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2015-10-09 22:27:29 UTC |
|---|
| Update date | 2017-01-19 02:36:10 UTC |
|---|
| FoodComEx ID | PC000027 |
|---|
| FoodDB Record | FDB001224 |
|---|
| Chemical Information |
|---|
| Name | L-Ascorbic acid |
|---|
| Description | Ascorbic acid, also known as acide ascorbique or L-ascorbate, belongs to the class of organic compounds known as butenolides. These are dihydrofurans with a carbonyl group at the C2 carbon atom. Ascorbic acid is a drug which is used to treat vitamin c deficiency, scurvy, delayed wound and bone healing, urine acidification, and in general as an antioxidant. it has also been suggested to be an effective antiviral agent. Ascorbic acid is an extremely weak basic (essentially neutral) compound (based on its pKa). dopamine and ascorbic acid can be converted into norepinephrine and dehydroascorbic acid through the action of the enzyme dopamine beta-hydroxylase. In humans, ascorbic acid is involved in the metabolic disorder called tyrosinemia, transient, of the newborn. Ascorbic acid is a very mild and grassy tasting compound. Outside of the human body, Ascorbic acid is found, on average, in the highest concentration within a few different foods, such as acerola, pepper (c. frutescens), and orange bell peppers and in a lower concentration in yogurts, yardlong beans, and peanuts. Ascorbic acid has also been detected, but not quantified in, several different foods, such as deerberries, cetacea (dolphin, porpoise, whale), acorns, lemon balms, and gelatins. This could make ascorbic acid a potential biomarker for the consumption of these foods. The L-enantiomer of ascorbic acid and conjugate acid of L-ascorbate. |
|---|
| CAS Number | 50-81-7 |
|---|
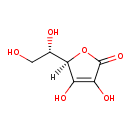
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| (+)-Ascorbate | biospider | | (+)-Ascorbic acid | biospider | | 3-Keto-L-gulofuranolactone | biospider | | 3-Oxo-L-gulofuranolactone | biospider | | Adenex | biospider | | Allercorb | biospider | | Antiscorbic vitamin | biospider | | Antiscorbutic vitamin | biospider | | Ascoltin | biospider | | Ascorbicap | db_source | | Ascorbicin | biospider | | Ascorin | biospider | | Ascorteal | biospider | | Ascorvit | biospider | | Cantan | biospider | | Cantaxin | biospider | | Catavin C | biospider | | Cebicure | biospider | | Cebion | biospider | | Cebione | db_source | | Cecon | biospider | | Cegiolan | biospider | | Ceglion | biospider | | Ceklin | biospider | | Celaskon | db_source | | Celin | biospider | | Cell C | biospider | | Cemagyl | biospider | | Cenetone | biospider | | Cereon | biospider | | Cergona | biospider | | Cescorbat | biospider | | Cetamid | biospider | | Cetane-Caps TC | biospider | | Cetebe | biospider | | Cetemican | biospider | | Cevalin | db_source | | Cevatine | biospider | | Cevimin | biospider | | Cevital | db_source | | Cevitamate | biospider | | Cevitamic acid | biospider | | Cevitamin | biospider | | Cevitan | biospider | | Cevitex | biospider | | Cewin | biospider | | Ciamin | biospider | | Cipca | biospider | | Citrovit | db_source | | Colascor | biospider | | Concemin | biospider | | E300 | db_source | | gamma-lactone L-threo-Hex-2-enonate | biospider | | gamma-lactone L-threo-Hex-2-enonic acid | biospider | | Hicee | biospider | | Hybrin | biospider | | Juvamine | biospider | | L-(+)-ascorbate | biospider | | L-(+)-ascorbic acid | biospider | | L-Ascorbate | biospider | | L-Ascorbic acid (8CI,9CI) | biospider | | L-threo-ascorbic acid | biospider | | Laroscorbine | biospider | | Lemascorb | biospider | | Proscorbin | biospider | | Redoxon | biospider | | Testascorbic | biospider | | Vicelat | biospider | | Vicin | biospider | | Viforcit | biospider | | Viscorin | biospider | | Vitace | biospider | | Vitacee | biospider | | Vitacimin | db_source | | Vitacin | biospider | | Vitamin C | db_source | | Vitamisin | biospider | | Vitascorbol | db_source | | Xitix | biospider |
|
|---|
| Chemical Formula | C6H8O6 |
|---|
| IUPAC name | (5R)-5-[(1S)-1,2-dihydroxyethyl]-3,4-dihydroxy-2,5-dihydrofuran-2-one |
|---|
| InChI Identifier | InChI=1S/C6H8O6/c7-1-2(8)5-3(9)4(10)6(11)12-5/h2,5,7-10H,1H2/t2-,5+/m0/s1 |
|---|
| InChI Key | CIWBSHSKHKDKBQ-JLAZNSOCSA-N |
|---|
| Isomeric SMILES | [H][C@@]1(OC(=O)C(O)=C1O)[C@@H](O)CO |
|---|
| Average Molecular Weight | 176.1241 |
|---|
| Monoisotopic Molecular Weight | 176.032087988 |
|---|
| Chemical Taxonomy |
|---|
| Description | Belongs to the class of organic compounds known as butenolides. These are dihydrofurans with a carbonyl group at the C2 carbon atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Dihydrofurans |
|---|
| Sub Class | Furanones |
|---|
| Direct Parent | Butenolides |
|---|
| Alternative Parents | |
|---|
| Substituents | - 2-furanone
- Vinylogous acid
- Alpha,beta-unsaturated carboxylic ester
- Enoate ester
- 1,2-diol
- Carboxylic acid ester
- Enediol
- Secondary alcohol
- Lactone
- Carboxylic acid derivative
- Oxacycle
- Monocarboxylic acid or derivatives
- Alcohol
- Hydrocarbon derivative
- Organic oxygen compound
- Carbonyl group
- Organic oxide
- Primary alcohol
- Organooxygen compound
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | -1.85 | AVDEEF,A (1997) |
|---|
| Experimental Water Solubility | 400 mg/mL at 40 oC | MERCK INDEX (1996) |
|---|
| Melting Point | Mp 190-192° | DFC |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | commercial |
|---|
| Production Method Reference | Not Available |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | solid |
|---|
| Storage Conditions | -80°C |
|---|
| Stability | Not Available |
|---|
| Purity | Not Available |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | Not Available |
|---|
| Provider Information |
|---|
|
| Commercial Vendors |
|---|
| AKSci | C510 |
|---|
| Cayman Chemical | 14656 |
|---|
| Glentham | GV5017 |
|---|
| Glentham | GV9871 |
|---|
| MetaSci | HMDB0000044 |
|---|
| Sigma-Aldrich | HMDB0000044 |
|---|
| Toronto Research Chemicals | A786990 |
|---|