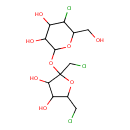
| Description | Noncalorific sweetener with good taste props.
One report suggests sucralose is a possible trigger for some migraine patients. Another study published in the Journal of Mutation Research linked doses of sucralose equivalent to 11,450 packets per day in a person to DNA damage in mice.; Results from over 100 animal and clinical studies in the FDA approval process unanimously indicated a lack of risk associated with sucralose intake. However, some adverse effects were seen at doses that significantly exceeded the estimated daily intake (EDI), which is 1.1 mg/kg/day. When the EDI is compared to the intake at which adverse effects are seen, known as the highest no adverse effects limit (HNEL), at 1500 mg/kg/day, there is a large margin of safety. The bulk of sucralose ingested is not absorbed by the gastrointestinal (GI) tract and is directly excreted in the feces, while 11-27% of it is absorbed. The amount that is absorbed from the GI tract is largely removed from the blood stream by the kidneys and eliminated in the urine with 20-30% of the absorbed sucralose being metabolized.; Sucralose belongs to a class of compounds known as organochlorides (or chlorocarbons). Some organochlorides, particularly those that accumulate in fatty tissues, are toxic to plants or animals, including humans. Sucralose, however, is not known to be toxic in small quantities and is extremely insoluble in fat; it cannot accumulate in fat like chlorinated hydrocarbons. In addition, sucralose does not break down or dechlorinate.; Sucralose can be found in more than 4,500 food and beverage products. It is used because it is a no-calorie sweetener, does not promote dental caries, and is safe for consumption by diabetics. Sucralose is used as a replacement for, or in combination with, other artificial or natural sweeteners such as aspartame, acesulfame potassium or high-fructose corn syrup. Sucralose is used in products such as candy, breakfast bars and soft drinks. It is also used in canned fruits wherein water and sucralose take the place of much higher calorie corn syrup based additives. Sucralose mixed with maltodextrin or dextrose (both made from corn) as bulking agents is sold internationally by McNeil Nutritionals under the Splenda brand name. In the United States and Canada, this blend is increasingly found in restaurants, including McDonald's, Tim Hortons and Starbucks, in yellow packets, in contrast to the blue packets commonly used by aspartame and the pink packets used by those containing saccharin sweeteners; though in Canada yellow packets are also associated with the SugarTwin brand of cyclamate sweetener.; Sucralose is a highly heat-stable artificial sweetener, allowing it to be used in many recipes with little or no sugar. Sucralose is available in a granulated form that allows for same-volume substitution with sugar. This mix of granulated sucralose includes fillers, all of which rapidly dissolve in liquids.[citation needed] Unlike sucrose which dissolves to a clear state, sucralose suspension in clear liquids such as water results in a cloudy state. For example, gelatin and fruit preserves made with sucrose have a satiny, near jewel-like appearance, whereas the same products made with sucralose (whether cooked or not) appear translucent and marginally glistening.[citation needed] While the granulated sucralose provides apparent volume-for-volume sweetness, the texture in baked products may be noticeably different. Sucralose is non-hygroscopic, meaning it does not attract moisture, which can lead to baked goods that are noticeably drier and manifesting a less dense texture than baked products made with sucrose. Unlike sucrose which melts when baked at high temperatures, sucralose maintains its granular structure when subjected to dry, high heat (e.g., in a 350 °F (177 °C) oven). Thus, in some baking recipes, such as crème brûlée, which require sugar sprinkled on top to partially or fully melt and crystallize, substituting sucralose will not result in the same surface texture, crispness, or crystalline structure.; Sucralose is a zero-calorie sugar substitute artificial sweetener. In the European Union, it is also known under the E number (additive code) E955. Sucralose is approximately 600 times as sweet as sucrose (table sugar), twice as sweet as saccharin, and 3.3 times as sweet as aspartame. Unlike aspartame, it is stable under heat and over a broad range of pH conditions. Therefore, it can be used in baking or in products that require a longer shelf life. The commercial success of sucralose-based products stems from its favorable comparison to other low-calorie sweeteners in terms of taste, stability, and safety. |
|---|
| Synonyms | | Synonym | Source |
|---|
| 1,6-Dichloro-1,6-dideoxy-b-D-fructofuranosyl 4-chloro-4-deoxy-a-D-galactopyranoside, 9CI | db_source | | 1',4,6'-Trichlorogalactosucrose | biospider | | 4,1',6'-Trichloro-4,1',6'-trideoxy-galacto-sucrose | biospider | | 4,1',6'-Trichloro-4,1',6'-trideoxygalactosucrose | db_source | | Splenda | db_source | | Sucralose [ban] | biospider | | Trichlorogalacto-sucrose | biospider | | Trichlorogalactosucrose | db_source | | Trichlorosucrose | biospider |
|
|---|