| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2017-11-02 09:49:22 UTC |
|---|
| Update date | 2017-11-02 09:49:22 UTC |
|---|
| FoodComEx ID | PC001050 |
|---|
| FoodDB Record | Not Available |
|---|
| Chemical Information |
|---|
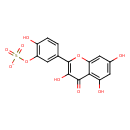
| Name | quercetin-3´-O-sulfate |
|---|
| Description | sulfate of the flavonol quercetin |
|---|
| CAS Number | Not Available |
|---|
| Structure | |
|---|
| Synonyms | |
|---|
| Chemical Formula | Not Available |
|---|
| IUPAC name | Not Available |
|---|
| InChI Identifier | InChI=1S/C15H10O10S/c16-7-4-9(18)12-11(5-7)24-15(14(20)13(12)19)6-1-2-8(17)10(3-6)25-26(21,22)23/h1-5,16-18,20H,(H,21,22,23)/p-1 |
|---|
| InChI Key | OSCLBBUATYLBQA-UHFFFAOYSA-M |
|---|
| Isomeric SMILES | OC1=CC(O)=C2C(=O)C(O)=C(OC2=C1)C1=CC(OS([O-])(=O)=O)=C(O)C=C1 |
|---|
| Average Molecular Weight | 381.29 |
|---|
| Monoisotopic Molecular Weight | 380.992191244 |
|---|
| Chemical Taxonomy |
|---|
| Class | flavonol |
|---|
| Family | Not Available |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| onions, apples.... |
| Production Data |
|---|
| Production Method | enzymatic synthesis |
|---|
| Production Method Reference | Valentová, K.; Káňová, K.; Di Meo, F.; Pelantová, H.; Chambers, C.; Rydlová, L.; Petrásková, L.; Křenková, A.; Cvačka, J.; Trouillas, P., et al. Chemoenzymatic preparation and biophysical properties of sulfated quercetin metabolites. Int. J. Mol. Sci. 2017, 18, 2231. |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | 1 to 10 mg |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | amorphous solid |
|---|
| Storage Conditions | RT |
|---|
| Stability | months |
|---|
| Purity | >95% |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | 1H NMR, 13C NMR, ESI-MS, LC-MS, UV |
|---|
| Provider Information |
|---|
| Contact Name | Contact Institution | Contact Email |
|---|
| Kateřina Valentová | Institute of Microbiology, Laboratory of Biotransformation, Vídeňská 1083, Prague, CZ, 14220 | kata.valentova@email.cz |
|