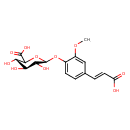| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation date | 2017-01-17 12:52:34 UTC |
|---|
| Update date | 2017-01-17 12:52:34 UTC |
|---|
| FoodComEx ID | PC001025 |
|---|
| FoodDB Record | Not Available |
|---|
| Chemical Information |
|---|
| Name | 4-O-beta-D-Glucuronyl-ferulic acid |
|---|
| Description | One of the main circulating metabolites of ferulic acid and its dietary derivatives
|
|---|
| CAS Number | Not Available |
|---|
| Structure | |
|---|
| Synonyms | | Synonym | Source |
|---|
| ferulic acid 4-O-beta-D-glucuronide | FoodComEx |
|
|---|
| Chemical Formula | Not Available |
|---|
| IUPAC name | Not Available |
|---|
| InChI Identifier | InChI=1S/C16H18O10/c1-24-9-6-7(3-5-10(17)18)2-4-8(9)25-16-13(21)11(19)12(20)14(26-16)15(22)23/h2-6,11-14,16,19-21H,1H3,(H,17,18)(H,22,23)/b5-3+/t11-,12-,13?,14?,16+/m0/s1 |
|---|
| InChI Key | TWSIWBHKRJLZCF-CNFYPOONSA-N |
|---|
| Isomeric SMILES | COC1=CC(\C=C\C(O)=O)=CC=C1O[C@@H]1OC([C@@H](O)[C@H](O)C1O)C(O)=O |
|---|
| Average Molecular Weight | 370.31 |
|---|
| Monoisotopic Molecular Weight | 370.08999678 |
|---|
| Chemical Taxonomy |
|---|
| Class | Hydroxycinnamic acid |
|---|
| Family | Not Available |
|---|
| Physico-Chemical Properties - Experimental |
|---|
| Property | Value | Reference |
|---|
| Experimental logP | Not Available | |
|---|
| Experimental Water Solubility | Not Available | |
|---|
| Melting Point | Not Available | |
|---|
|
| Foods of Origin |
|---|
| Food | Content Range | Average | Reference |
|---|
| Food | | | Reference |
|---|
|
| Production Data |
|---|
| Production Method | chemical synthesis |
|---|
| Production Method Reference | S. Galland, N. Rakotomanomana, C. Dufour, N. Mora, O. Dangles, Org. Biomol. Chem. 2008, 6, 4253-4260. |
|---|
| Production Method Reference File | Not Available |
|---|
| Quantity Available | Production upon request, in g |
|---|
| Delivery Time | Not Available |
|---|
| Storage Form | powder |
|---|
| Storage Conditions | -18°C |
|---|
| Stability | High |
|---|
| Purity | >90% |
|---|
| Spectra |
|---|
| Spectral Data Upon Request | 1H NMR, 13C NMR |
|---|
| Provider Information |
|---|
| Contact Name | Contact Institution | Contact Email |
|---|
| Olivier Dangles | University of Avignon, Sécurité et Qualité des Produits d'Origine Végétale, Campus JH Fabre 301 rue Baruch de Spinoza, BP 21239, Cedex 9,, Avignon , FR, 84916 | olivier.dangles@univ-avignon.fr |
|